Ferro-aging: A Novel Mechanism where Vitamin C Targets ACSL4 to Alleviate Primate Aging


Introduction
Aging is a progressive process marked by the functional decline of multiple organs, with oxidative stress serving as a central driver; however, specific targetable pathways have remained elusive. A study published in Cell Metabolism by Liu Guanghui's team from the Institute of Zoology, Chinese Academy of Sciences, defines for the first time a novel aging axis —— "ferro-aging," and confirms that ACSL4 is its core execution factor. Vitamin C (VC) can directly bind to and inhibit ACSL4, thereby blocking ferro-aging. Long-term VC supplementation in non-human primates has been shown to delay aging, improve metabolic and neurological functions, and reverse biological age, providing a new theoretical framework and targets for anti-aging interventions.

Research Background
Dysregulation of iron metabolism is closely associated with aging, yet its causal role in primates has not been fully established. ACSL4 increases the sensitivity of cell membranes to iron-driven lipid peroxidation, but its specific function in the context of aging requires further elucidation. Traditional antioxidants have shown inconsistent results; as a classic antioxidant, Vitamin C's molecular mechanisms in anti-aging and its long-term effects in primates lack systematic validation.
Research Objectives
To define the ferro-aging pathway in primate aging and clarify the central role of ACSL4; to identify inhibitors of ferro-aging and elucidate their mechanisms; and to validate the anti-aging efficacy of targeting ferro-aging in non-human primates while assessing its impact on biological age.
Research Methods
Clinical Sample & Database Analysis:
Collection of human multi-organ tissues and serum to analyze the correlation between iron metabolism, ACSL4, and aging; construction of ferro-aging gene sets and multi-dimensional aging clocks.
Animal Models:
Establishment of normal aging, iron overload, and progeria mouse models, with ACSL4 knockout performed via CRISPR-Cas9; long-term (40-month) VC intervention in cynomolgus monkeys to evaluate multi-organ functions.
Cellular Experiments:
Construction of various cellular senescence models, inducing ferro-aging via iron ions and erastin (a ferroptosis inducer) to verify the regulatory role of ACSL4 in senescence and the intervention effects of VC.
Molecular Mechanism Validation:
Identification of direct VC targets through Pull-down assays combined with mass spectrometry; validation of VC's inhibitory effect on ACSL4 using enzyme activity assays and molecular docking; detection of changes in lipid metabolism and the Nrf2 pathway.
Multi-dimensional Aging Assessment:
Quantification of biological age using multi-omics aging clocks, and assessment of VC's systemic effects via MRI and serum biochemical indicators.
Research Workflow
- Validating the correlation between multi-tissue iron accumulation and lipid peroxidation in primate aging;
- Defining the central role of ACSL4 in mediating iron-dependent cellular senescence;
- Identifying VC as a direct inhibitor of ACSL4 and elucidating its molecular mechanism;
- Validating the anti-aging effects of targeting ACSL4/ferro-aging in both mice and non-human primates.
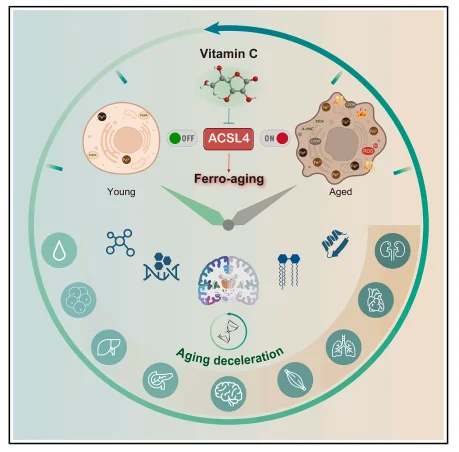
Graphical Abstract
Main Results
1. Iron Accumulation and Lipid Peroxidation are Core Hallmarks of Primate Aging
Iron deposition occurs across multiple tissues in humans and monkeys as they age, accompanied by the upregulation of ACSL4 and lipid peroxidation products (MDA, 4-HNE); ferro-aging gene sets are enriched in aged monkey tissues, particularly in metabolic tissues.
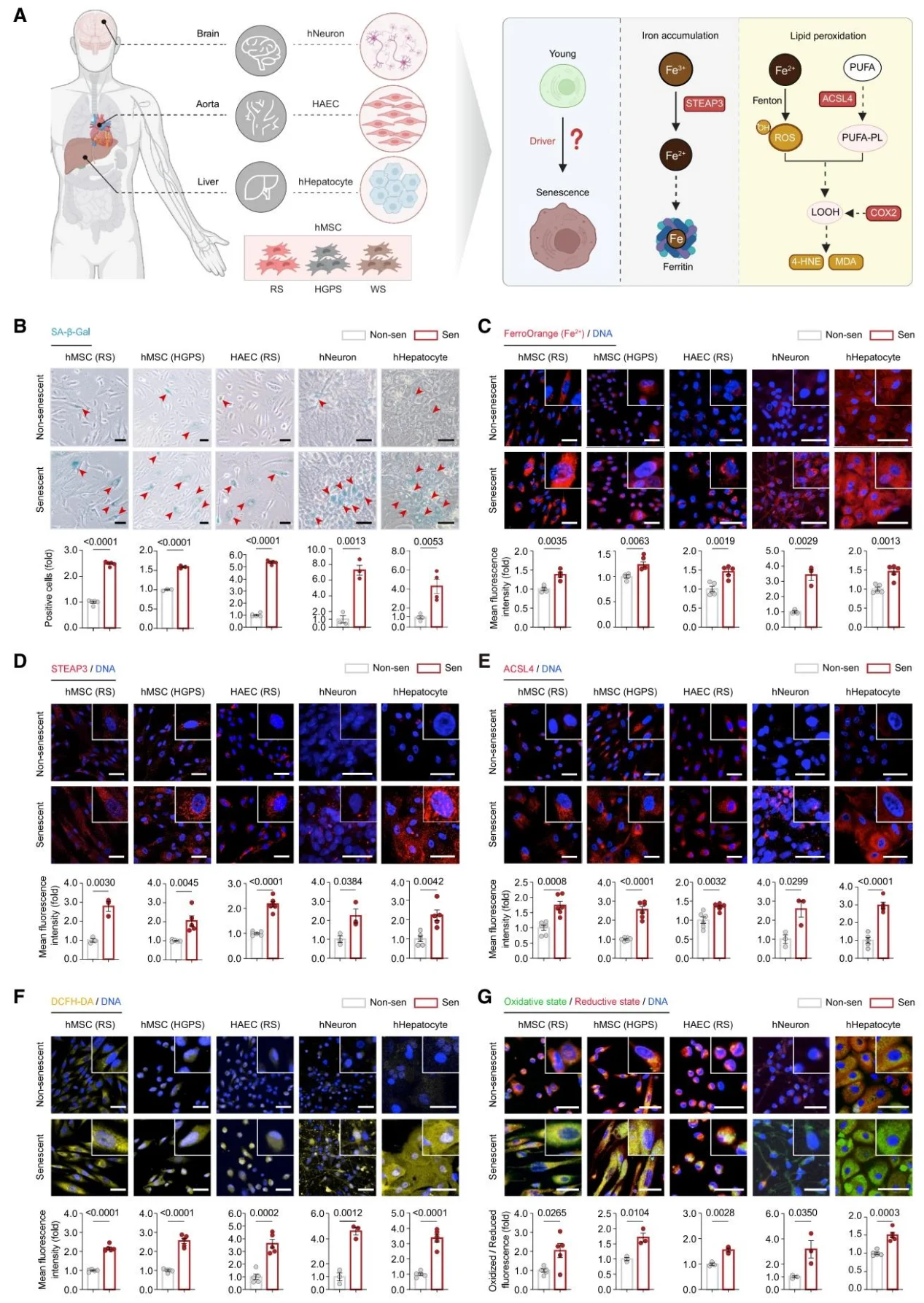
Figure 1. Dysregulation of Iron Homeostasis and Lipid Peroxidation Jointly Define Cellular Senescence.
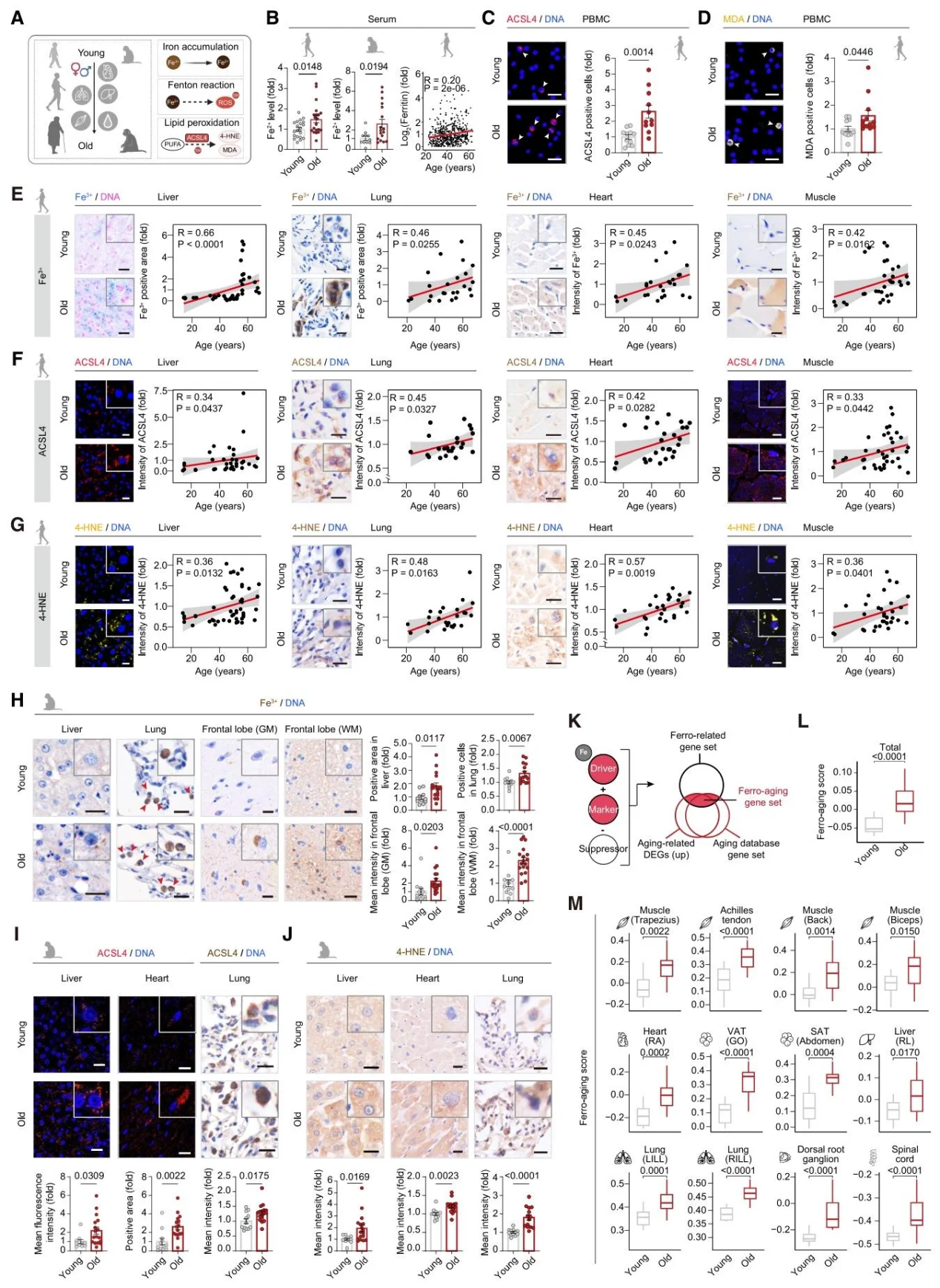
Figure 2. Ferro-aging Signatures in Aged Human and Primate Tissues.
2. ACSL4 is a Core Execution Factor of Ferro-aging
Iron overload induces the upregulation of ACSL4, triggering cellular senescence; overexpression of ACSL4 promotes aging, while knockdown inhibits the process. Target knockout of ACSL4 in mouse livers improves aging-related phenotypes and alleviates symptoms in progeria mouse models.
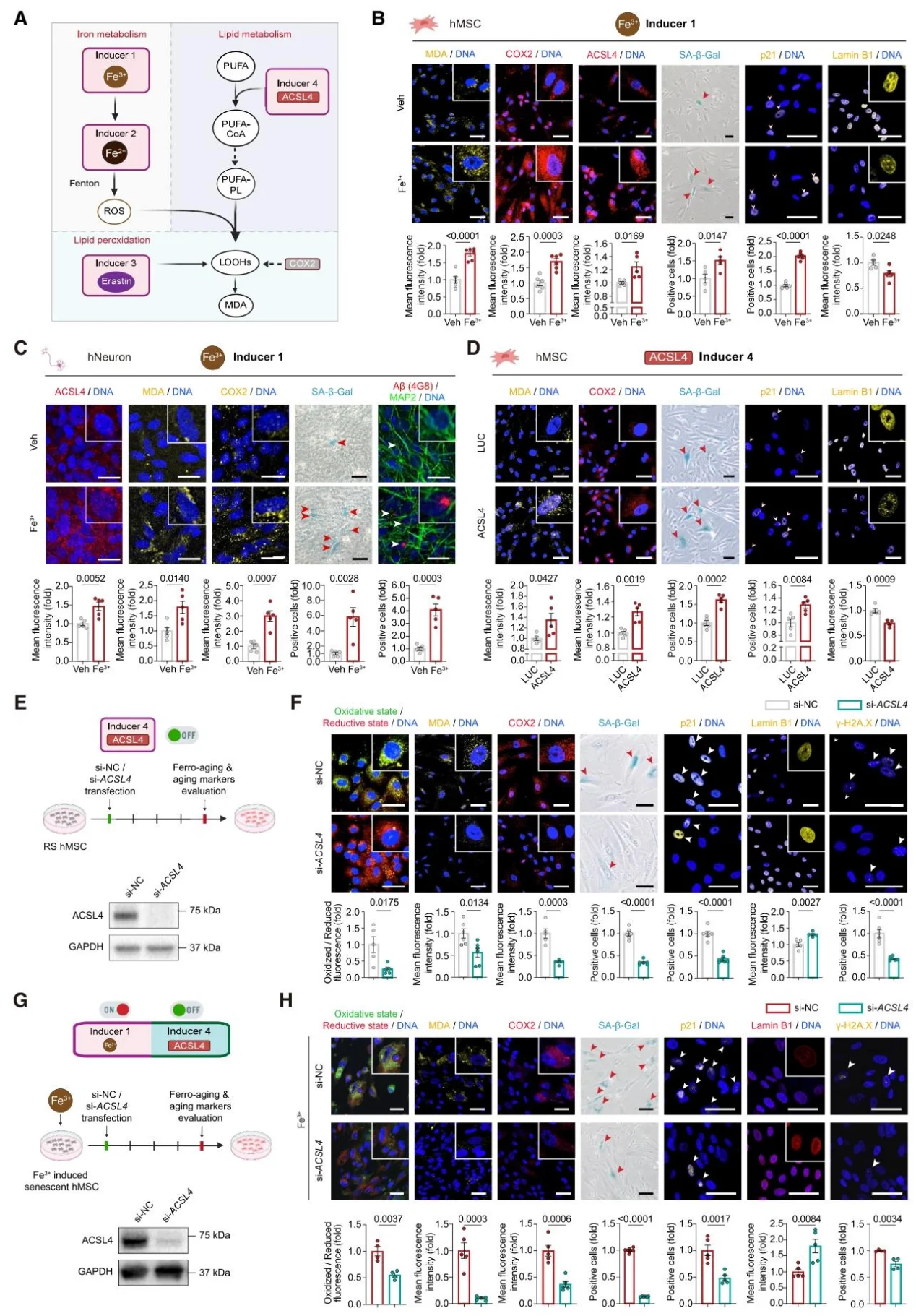
Figure 3. ACSL4 Mediates Iron-driven Senescence and Lipid Peroxidation.
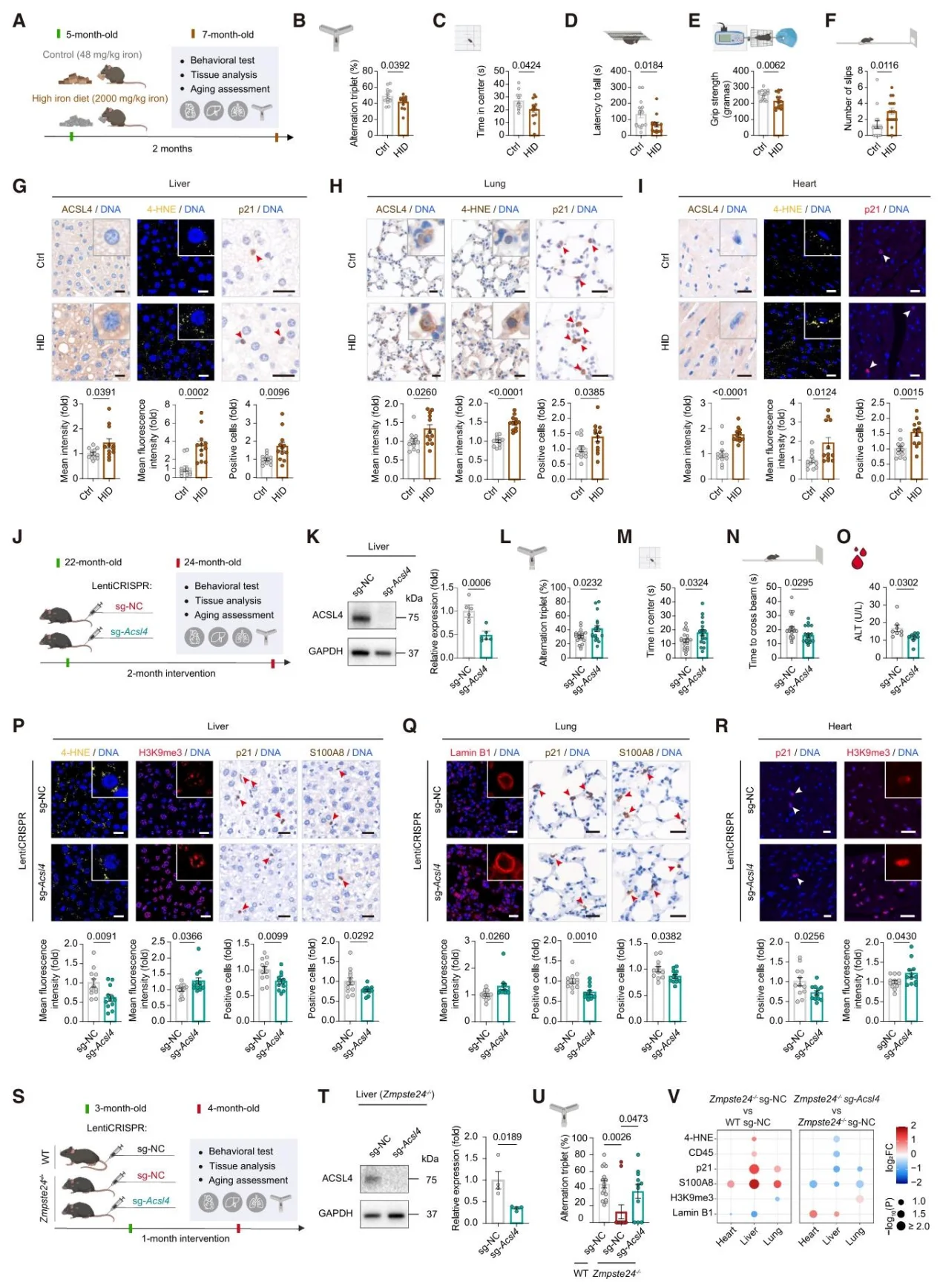
Figure 4. Liver-specific Genetic Targeting of ACSL4 Ameliorates Systemic Aging in Mice.
3. Vitamin C Directly Inhibits ACSL4
VC is a potent inhibitor of ferro-aging. Pull-down assays combined with mass spectrometry identified ACSL4 as its direct target; VC inhibits ACSL4 activity in a dose-dependent manner (binding sites: Thr278/Ser279/Thr469) while simultaneously activating the Nrf2 antioxidant pathway.
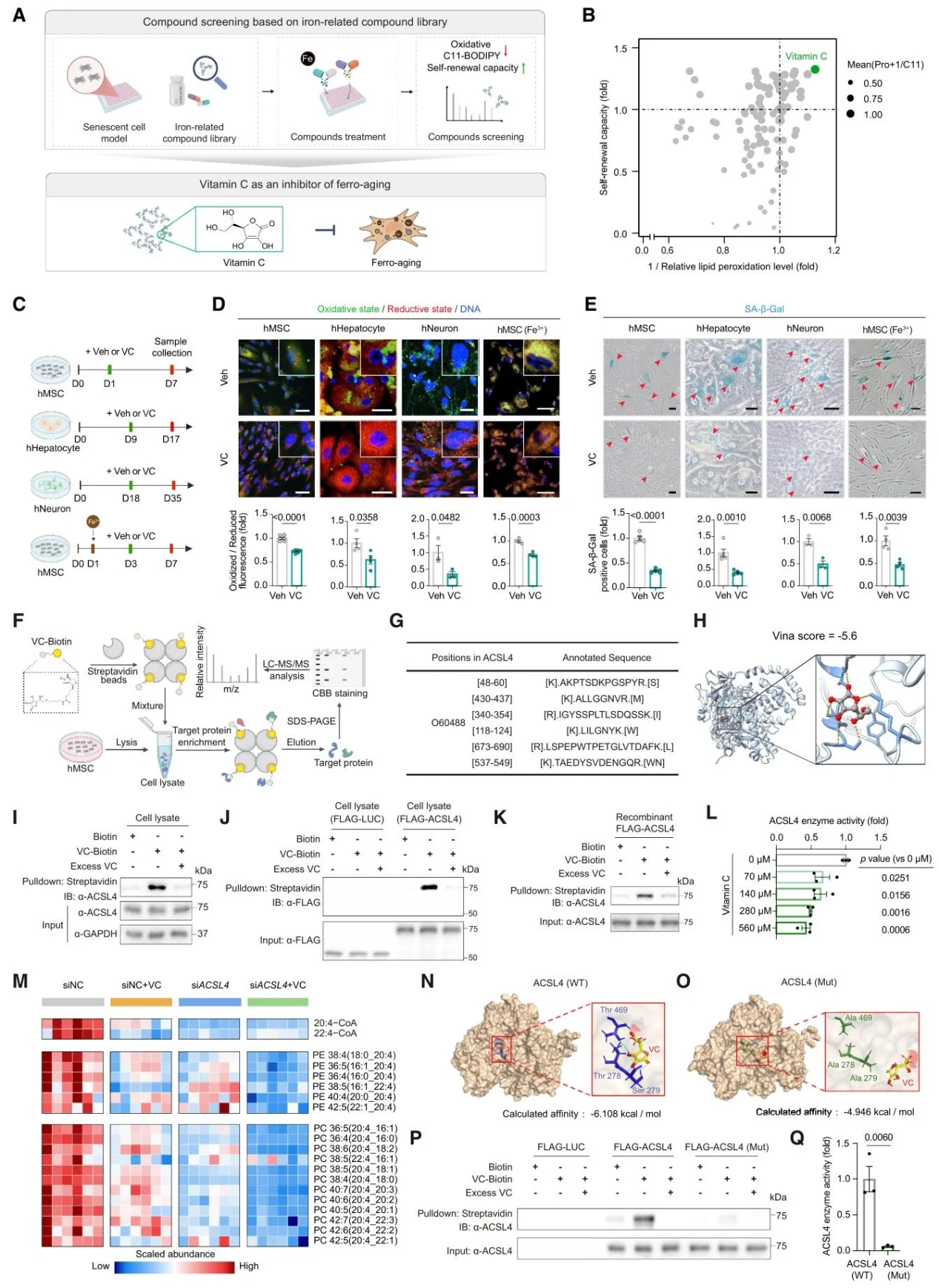
Figure 5. Vitamin C is a Direct Inhibitor of ACSL4.
4. Long-term VC Supplementation Delays Primate Aging
After long-term VC intervention in monkeys, ferro-aging scores across multiple tissues decreased and aging biomarkers were reduced; multi-omics clocks confirmed that it can reverse biological age, improve cortical structures, lipid profiles, and insulin resistance, and reduce visceral fat.
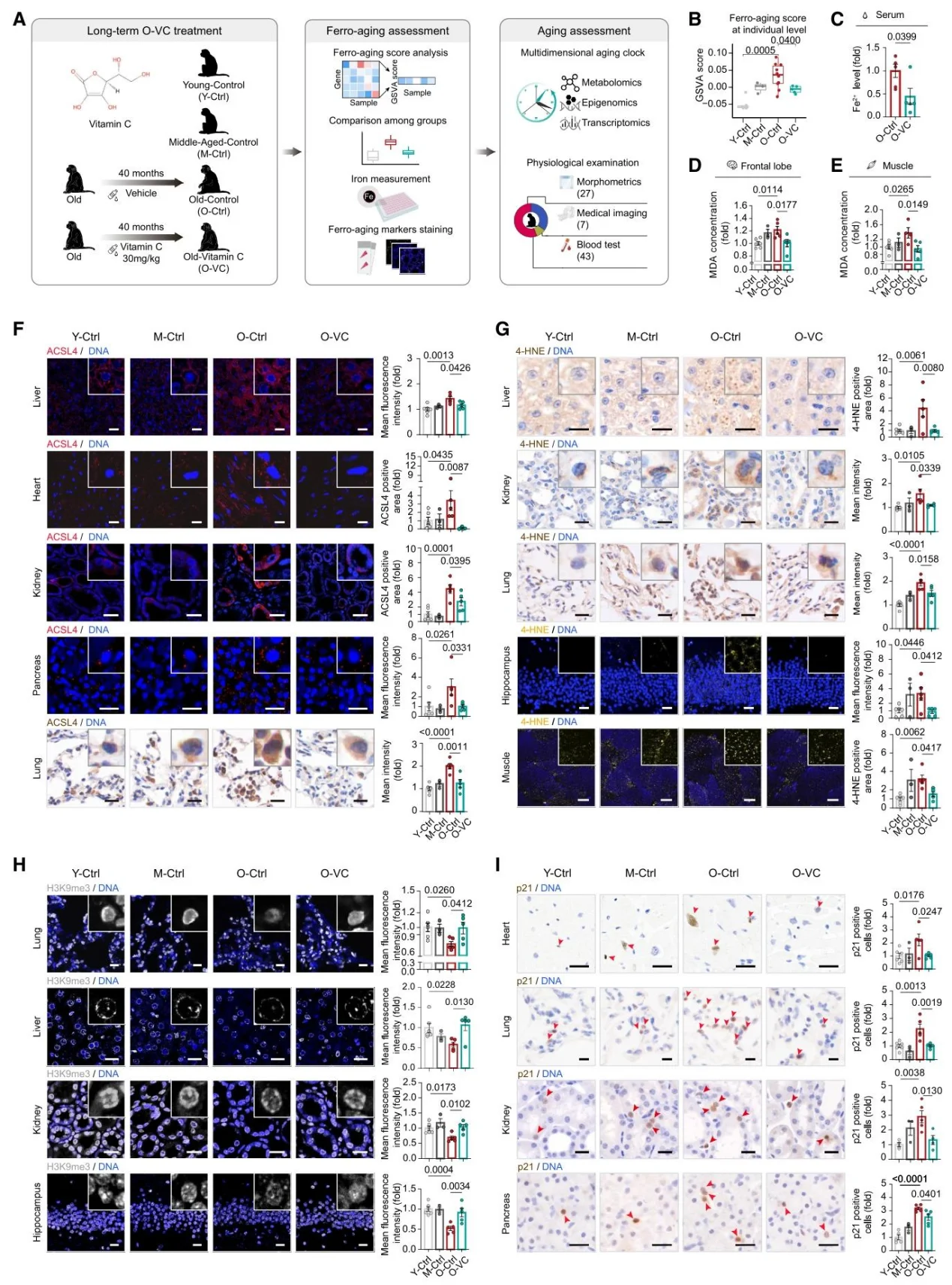
Figure 6. Long-term Vitamin C Supplementation Attenuates Ferro-aging in Non-human Primates.
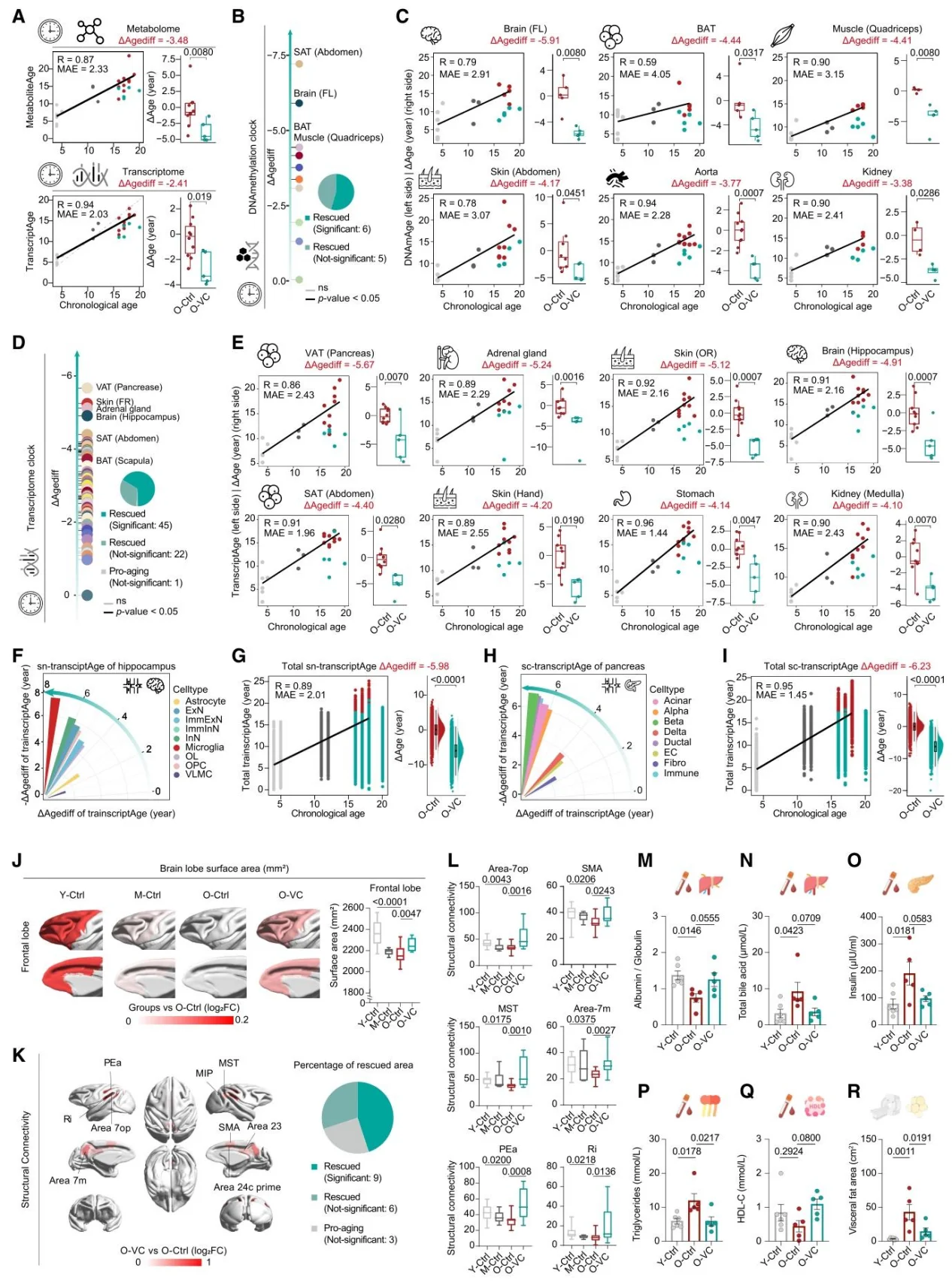
Figure 7. Vitamin C Exerts Multi-organ Anti-aging Effects and Reverses Biological Age.
Significance & Innovation
- Mechanistic Innovation: First to define ferro-aging as a central axis of primate aging, distinguishing it from acute ferroptosis and explaining the chronic, systemic characteristics of senescence.
- Target & Molecular Innovation: Identified ACSL4 as a key molecule in ferro-aging, with VC exerting a dual effect through the direct inhibition of ACSL4 and activation of Nrf2.
- Translational Innovation: Primate experiments confirmed that long-term VC intervention is safe and effective, providing a low-cost, high-safety translatable strategy for human anti-aging.
Summary
This study elucidates the central role of ferro-aging in primate senescence, identifying ACSL4 as a critical mediator of iron-dependent aging and demonstrating that VC blocks ferro-aging by directly inhibiting ACSL4. Non-human primate experiments verify that VC can delay multi-organ aging, reverse biological age, and improve neurological and metabolic functions. This provides theoretical support for anti-aging strategies targeting ferro-aging, positioning Vitamin C as a promising agent for clinical translation.
Ubigene consistently upholds the core philosophy of "Making Gene Editing Easier," continuously iterating its products and services. We have successfully completed over 13,000 gene-editing cases and offer 13,000+ cell products (including 10,000+ KO cells). Our independently developed innovative technologies have improved gene-editing efficiency by 10-20 times compared to traditional methods. To date, Ubigene has provided high-quality gene-editing services and products to over 10,000 life science laboratories, pharmaceutical companies, and CROs.
ACSL4 (Acyl-CoA Synthetase Long-Chain Family Member 4) is a key molecule regulating ferroptosis. It primarily influences the esterification of polyunsaturated fatty acids (PUFAs) in cell membranes, participating in the initiation and amplification of lipid peroxidation, thereby driving iron-dependent cell death. Its expression level is closely correlated with cellular sensitivity to ferroptosis. If you wish to conduct research on ACSL4, Ubigene can provide ACSL4 knockout cell lines, including HEK293, A549, and HeLa backgrounds. For personalized gene-editing needs, please feel free to consult us!
Contact us to learn more>>>


