Vascular STING Signaling Regulates NK Cell Infiltration in Small Cell Lung Cancer


Introduction
Small cell lung cancer (SCLC) often exhibits a "cold tumor" phenotype, characterized by sparse immune infiltration and poor responsiveness to immunotherapy. Neuroendocrine SCLC, due to defects in MHC-I expression, is expected to be susceptible to NK cell-mediated cytotoxicity; however, in clinical settings, NK cells are largely excluded from tumor regions. A study published in Cancer Cell by Marco Campisi and colleagues demonstrates for the first time that tumor vasculature represents a primary barrier to NK cell recruitment. Activation of vascular STING signaling restores NK cell trafficking and cytotoxic function, significantly enhancing the efficacy of DLL3-targeted CAR-NK therapy, thereby providing a new therapeutic direction for SCLC.

Research Background
The immunosuppressive tumor microenvironment (TME) of SCLC is a major driver of therapeutic resistance. Although the neuroendocrine subtype exhibits defects in MHC-I antigen presentation, insufficient NK cell infiltration prevents this potential immune advantage from being realized. Tumor vasculature serves as a physical barrier for immune cell entry into the TME, yet its regulatory role remains poorly understood. STING signaling is known to activate inflammatory responses; however, it is frequently silenced in SCLC tumor cells. Whether STING activity in vascular and stromal compartments can be therapeutically leveraged remains an open question. In addition, CAR-NK therapy in solid tumors is often limited by inadequate infiltration, highlighting the need for optimized combination strategies.
Research Objectives
- To identify the key mechanisms underlying insufficient NK cell infiltration in SCLC and to validate the regulatory role of tumor vasculature;
- To elucidate the impact of vascular STING activation on NK cell function;
- To develop a combinational therapeutic strategy integrating STING agonists with DLL3-targeted CAR-NK cells to improve immunotherapy outcomes in SCLC.
Research Methods
Clinical Sample Analysis:
Clinical samples from 34 SCLC patients were collected, and NK cell infiltration as well as vascular status were analyzed using multiplex immunofluorescence and spatial transcriptomics; public datasets were integrated to validate prognostic correlations.
Animal Models:
Syngeneic SCLC models and humanized vascularized xenograft models were established to evaluate the in vivo therapeutic efficacy of the combination therapy involving STING agonists and CAR-NK cells.
Cellular Experiments:
SCLC cell lines and primary NK cells were utilized to verify sensitivity via co-culture and cytotoxicity assays; CRISPR-Cas9 technology was employed to knock out key genes for mechanistic investigation; STING agonist treatment was used to detect the expression of vascular adhesion molecules and chemokines.
Molecular Mechanism Validation:
Signaling communication between vasculature and immune cells following STING activation was elucidated using DynaMITE-seq and CellChat analysis.
Therapeutic Validation:
DLL3-targeted CAR-NK cells were constructed to assess the potentiation effects of STING agonists in both in vitro and in vivo models.
Research Strategy
1. Theoretical Validation
Validating the paradox of MHC-I⁻ˡᵒʷ/ⁿᵉᵍ SCLC's in vitro sensitivity to NK cells versus its insufficient in vivo infiltration;
2. Identification of Key Barriers
Identifying tumor vasculature as the key barrier to NK cell infiltration;
3. Elucidation of Migration & Activation Mechanisms
Elucidating the mechanisms by which vascular STING activation promotes NK cell migration and activation;
4. Validation of Anti-SCLC Efficacy
Validating the anti-SCLC efficacy of the combination therapy involving STING agonists and CAR-NK cells.
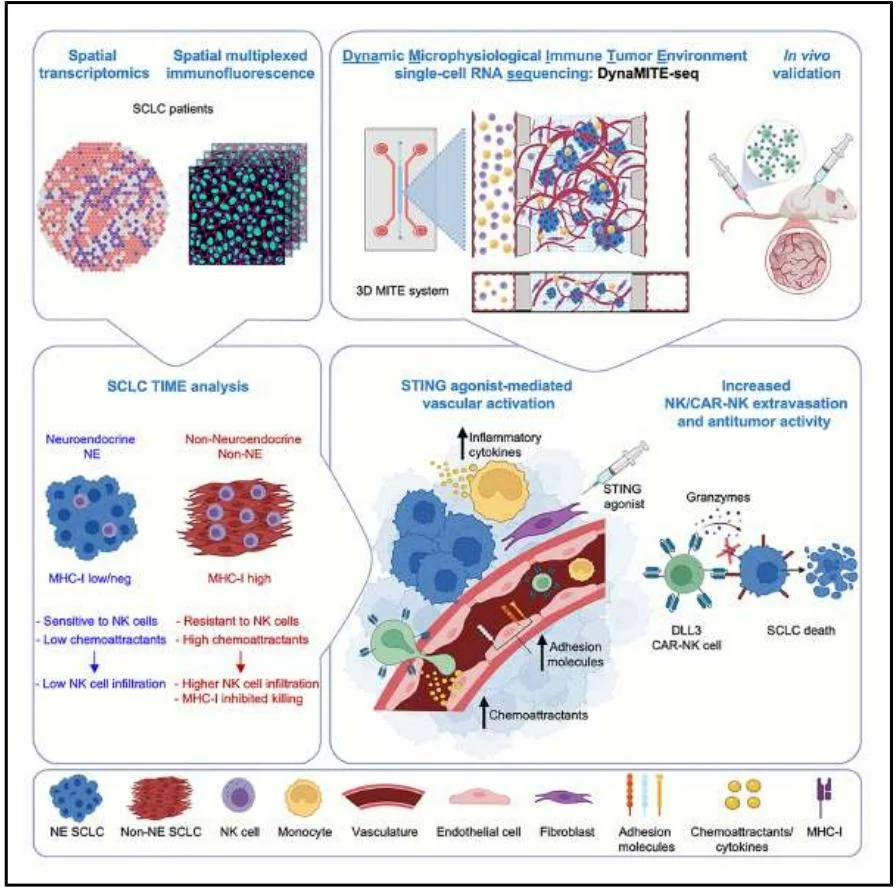
Graphical Abstract
Main Results
1. MHC-Iˡᵒʷ/neg SCLC is sensitive to NK cells in vitro but exhibits insufficient infiltration in vivo
MHC-Iˡᵒʷ/neg SCLC cell lines are highly sensitive to NK cell-mediated killing, yet NK cell infiltration is sparse within this subtype in patient samples. Furthermore, NK cells tend to localize in MHC-Ihigh regions, and patients with low NK infiltration exhibit shorter survival periods.
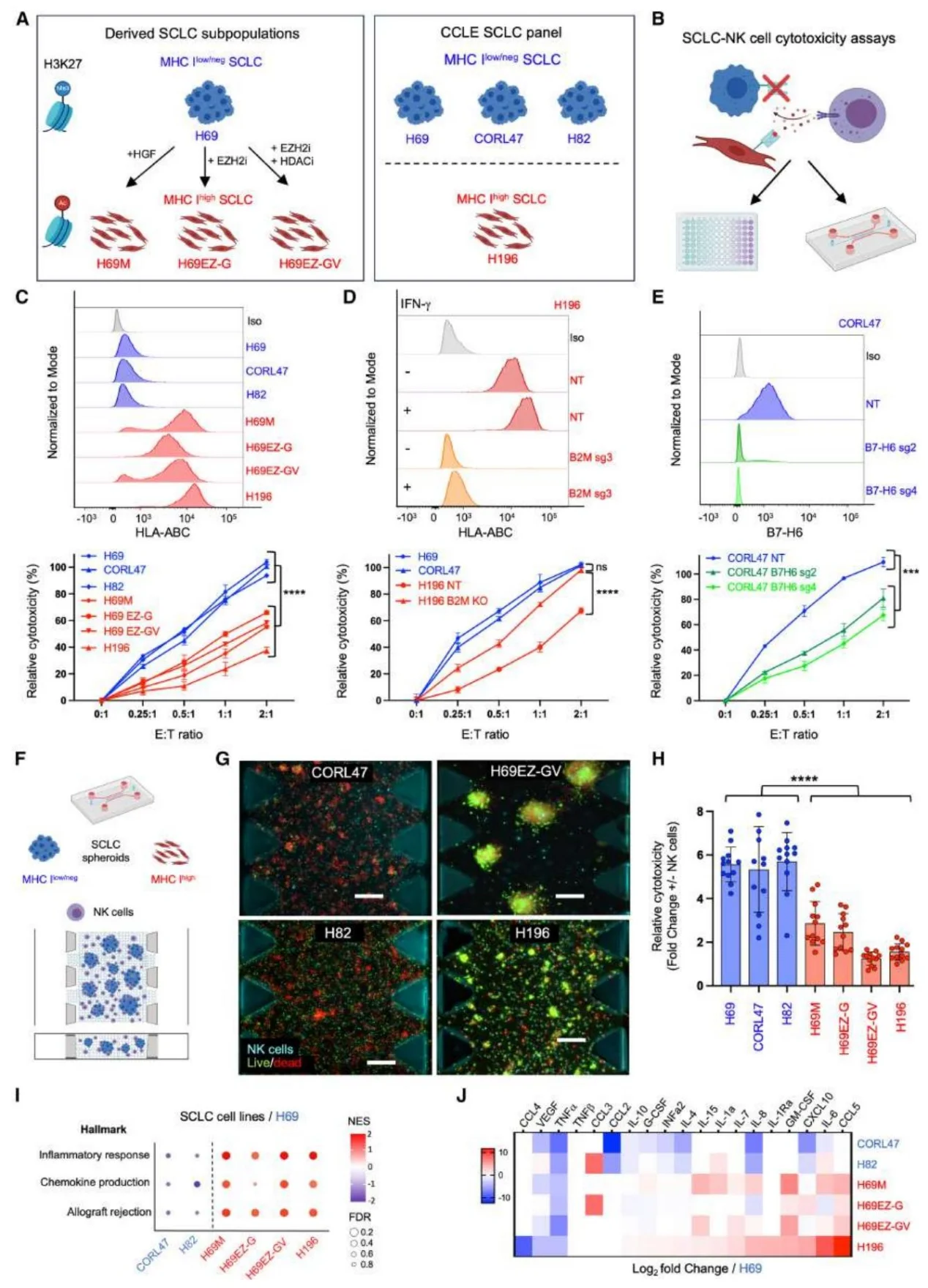
Figure 1. MHC Class I-low/negative Small Cell Lung Cancer (SCLC) is Sensitive to NK Cell-Mediated Cytotoxicity.
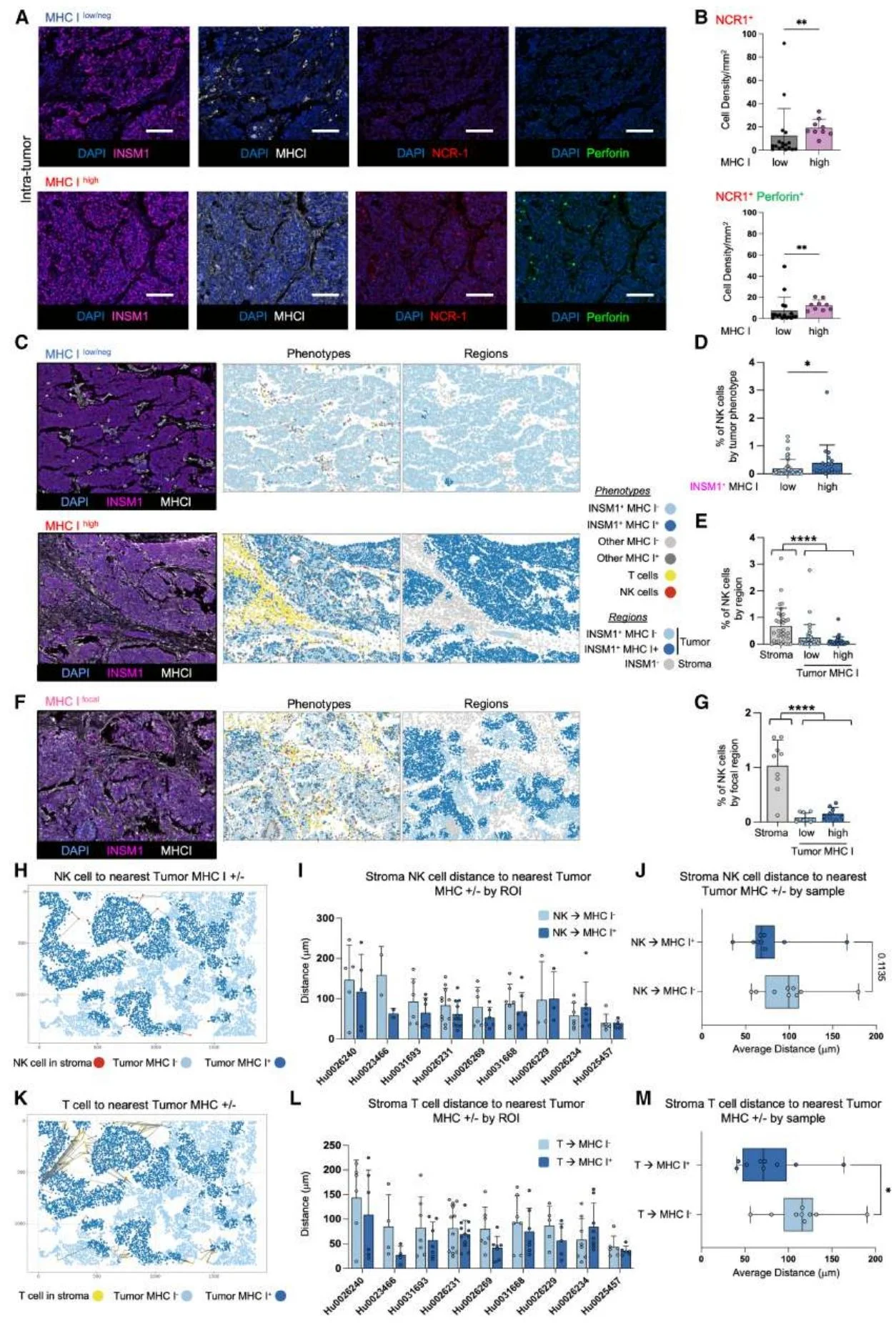
Figure 2. MHC-Iˡᵒʷ/ⁿᵉᵍ Small Cell Lung Cancer (SCLC) Exhibits Sparse and Regional NK Cell Infiltration.
2. Tumor vasculature is the core barrier to NK cell infiltration
The expression of chemokines is downregulated in MHC-Iˡᵒʷ/neg SCLC. STING is expressed at low levels in tumor cells but at high levels in vascular stromal cells. STING agonists can induce vascular endothelial cells to upregulate adhesion molecules (VCAM1, ICAM1) and chemokines (CXCL10); vasculature in MHC-Ihigh regions exhibits higher activation features and co-localizes with NK cells.
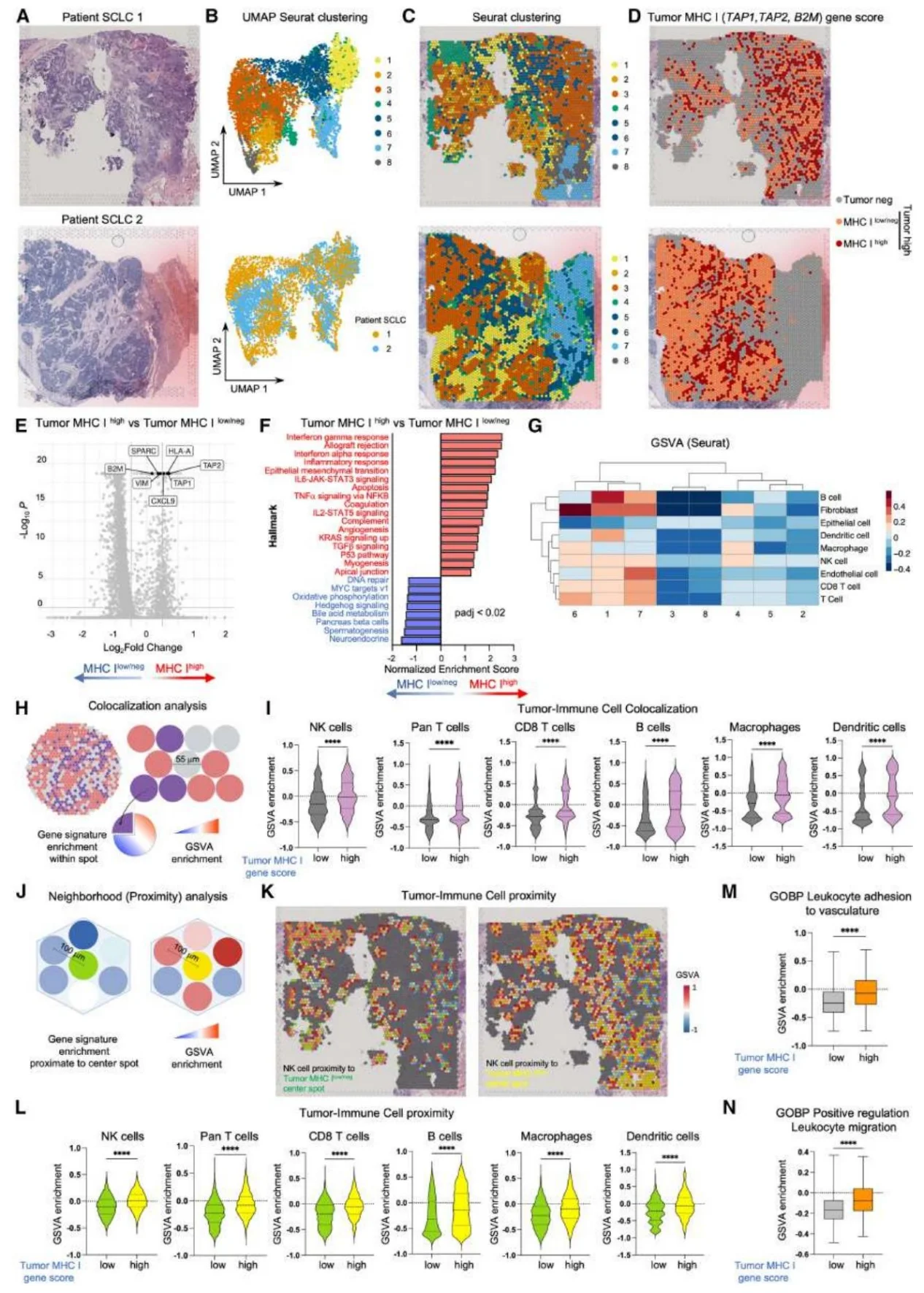
Figure 3. TIME Analysis of MHC-Iˡᵒʷ/ⁿᵉᵍ SCLC Reveals Insufficient Immune Engagement and Vascular Activation.
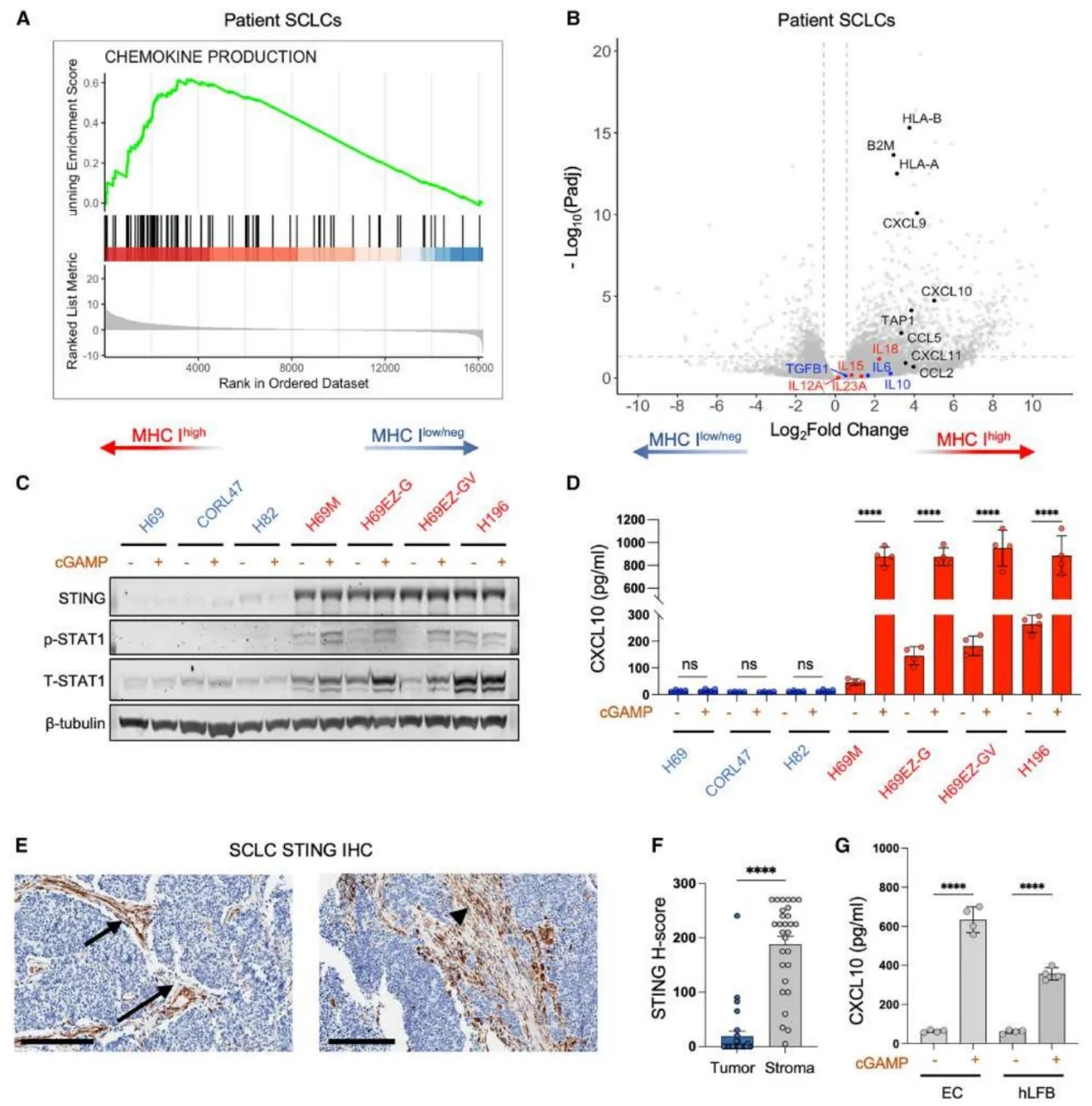
Figure 4. TIME Analysis of MHC-Iˡᵒʷ/ⁿᵉᵍ SCLC Shows Mismatch Between Intrinsic and Extrinsic Immune Features.
3. Vascular STING activation promotes NK cell migration and activation
STING agonists combined with monocytes can activate inflammatory and leukocyte extravasation pathways in the vascular endothelium, enhancing VCAM1 and SELE-mediated interactions between endothelial cells and NK cells. In this environment, NK cell activation markers are upregulated, inhibitory receptors are downregulated, and cytotoxicity is enhanced.
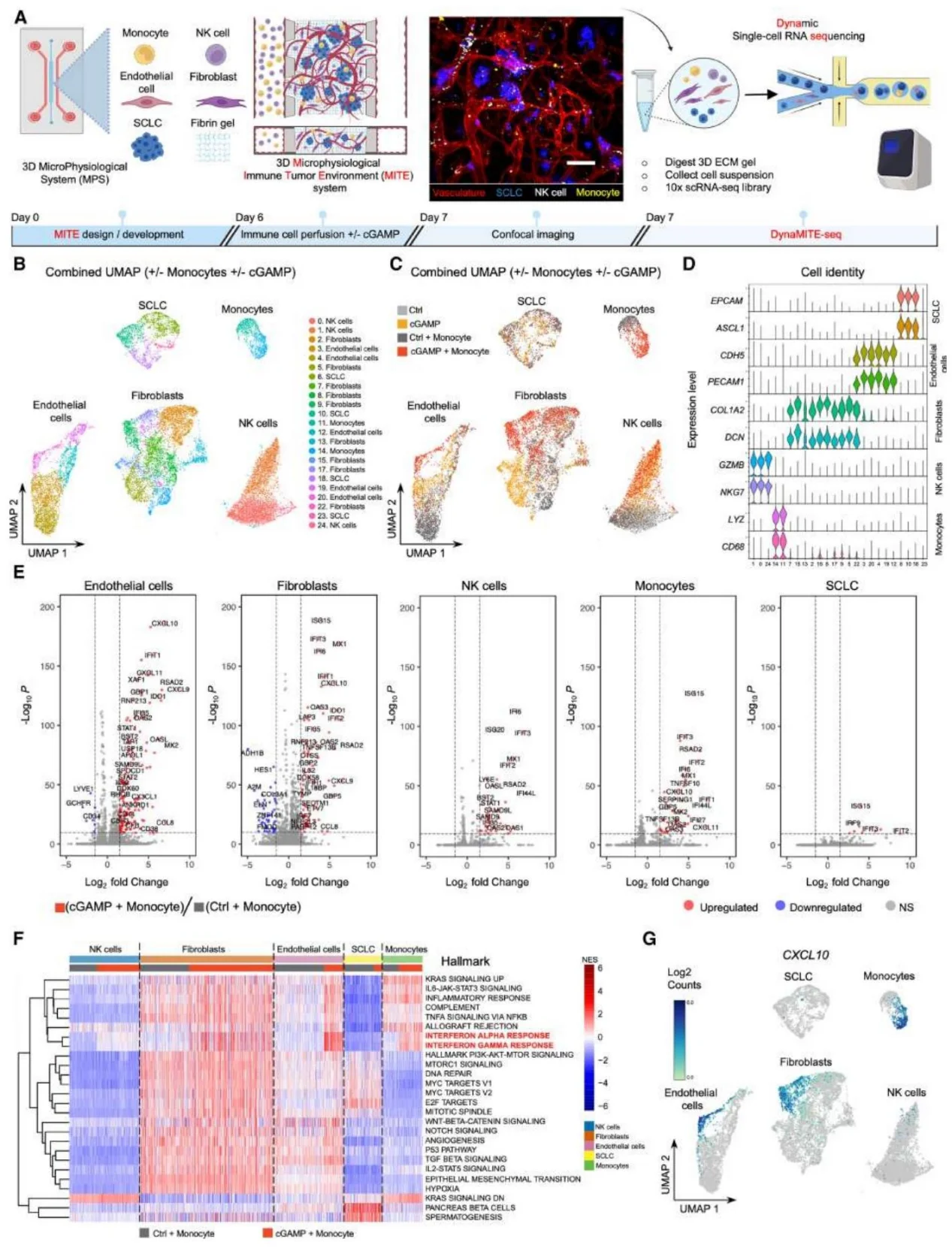
Figure 5. High-Resolution Characterization of a Perturbable Microphysiological Immune Tumor Environment (MITE).
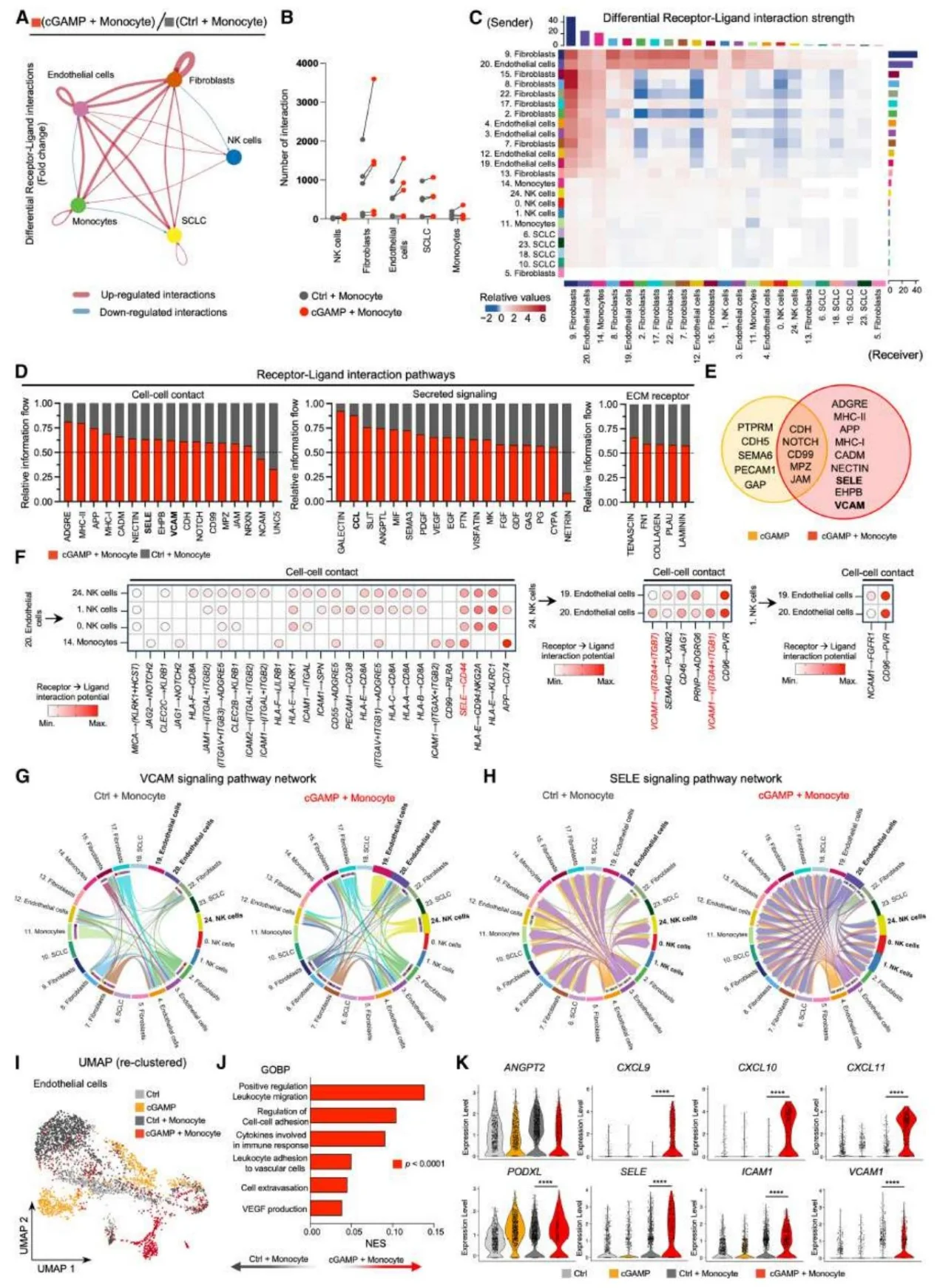
Figure 6. DynaMITE-seq and Receptor-Ligand Interaction Analysis Reveal STING Agonist-Mediated Vascular Activation.
4. STING agonists enhance the efficacy of DLL3 CAR-NK therapy
DLL3 CAR-NK cells demonstrate superior SCLC-killing capability compared to unmodified NK cells. STING agonists can increase the vascular adhesion and tumor infiltration of CAR-NK cells. In vivo experiments show that the combination treatment significantly inhibits tumor growth and increases intratumoral CAR-NK cell infiltration and inflammatory cytokine levels.
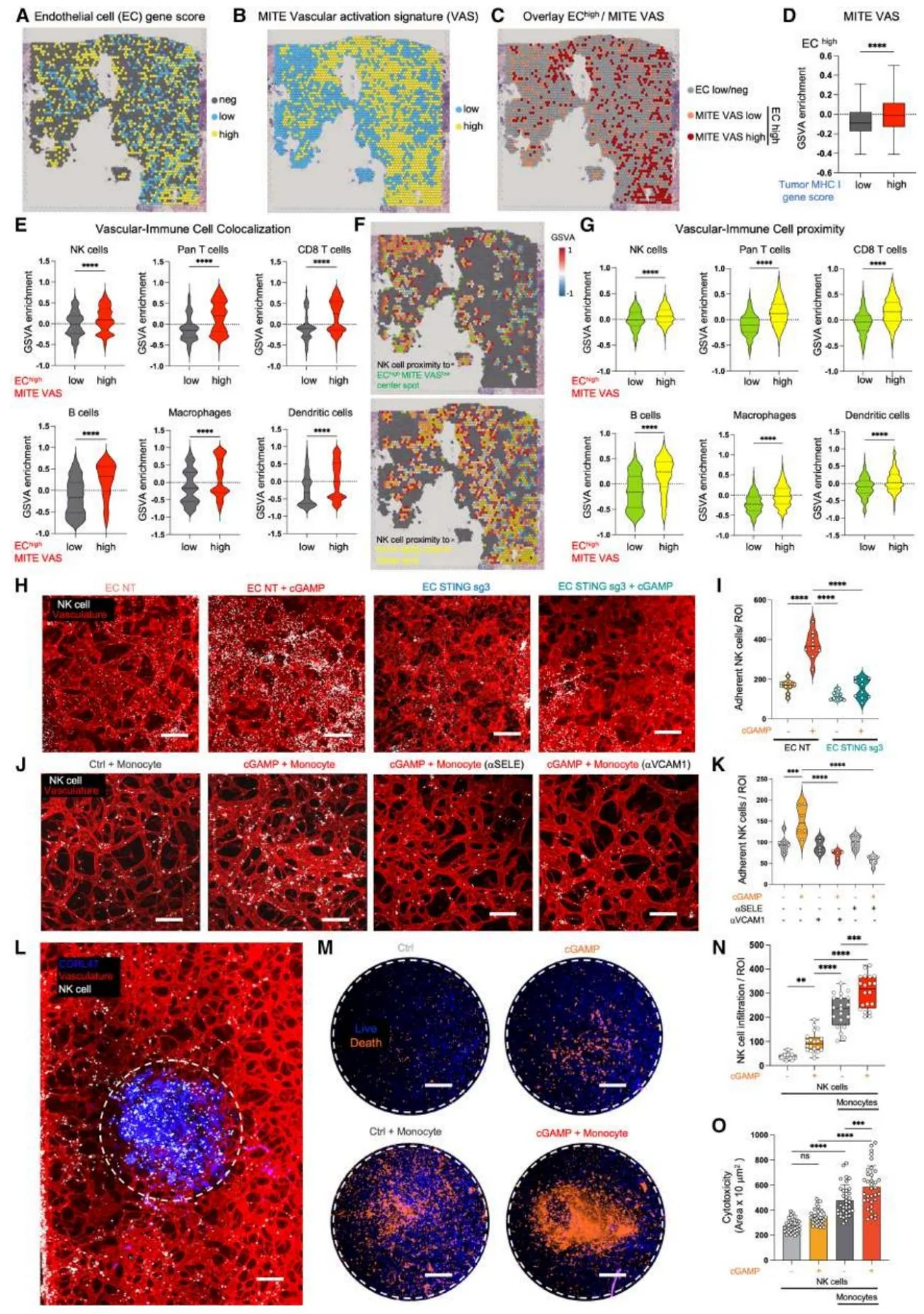
Figure 7. Integrated Spatial and DynaMITE-seq Analysis Indicates That STING Activation Facilitates NK Cell Extravasation and Enhances Anti-Tumor Activity in SCLC.
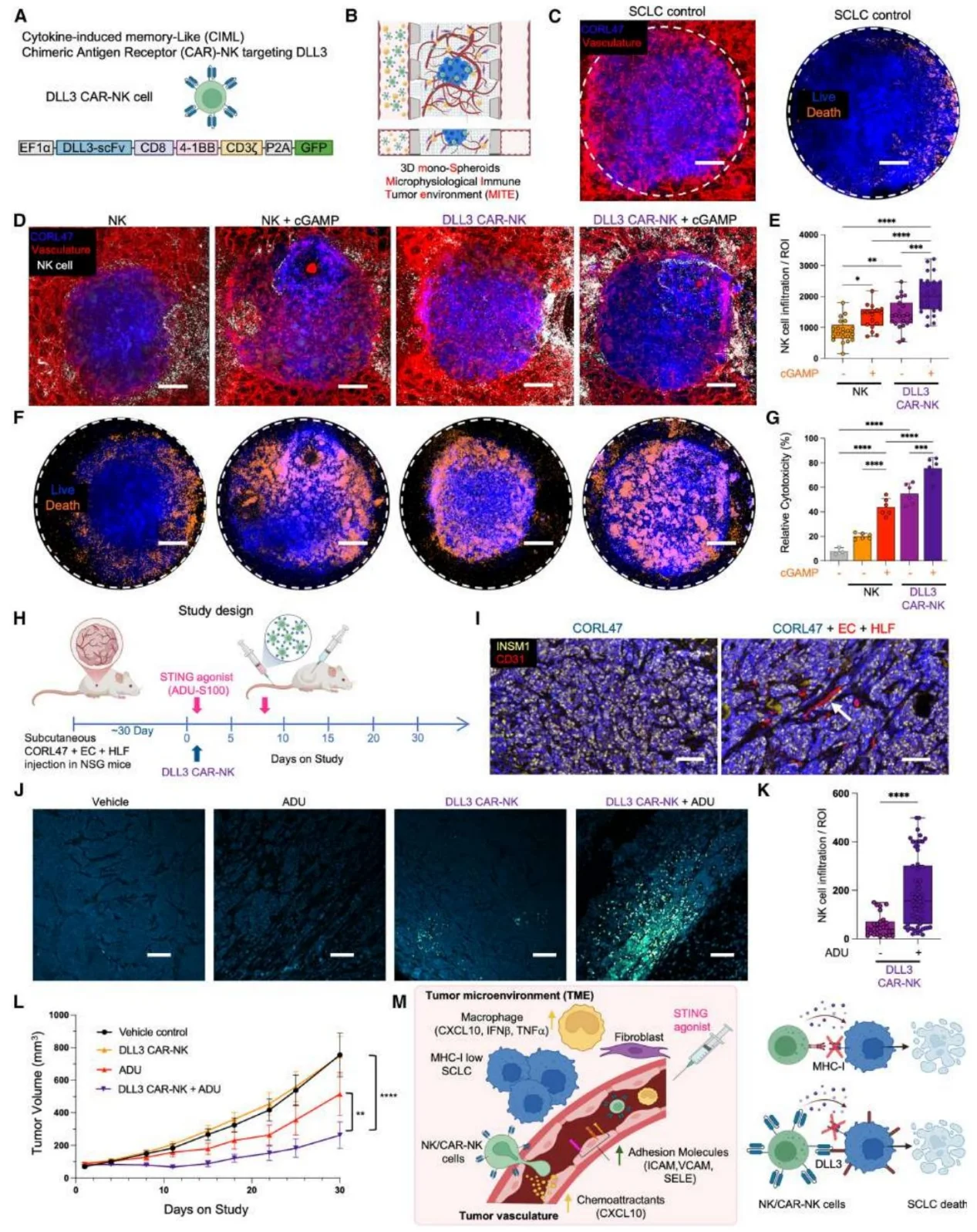
Figure 8. STING Agonism Facilitates DLL3 CAR-NK Cell Extravasation and Enhances Anti-Tumor Activity in a Humanized Vascularized MHC-Iˡᵒʷ/ⁿᵉᵍ SCLC Xenograft Model.
Significance & Innovation
- Mechanistic Innovation: Revealing tumor vasculature as the key barrier to NK cell infiltration in SCLC; elucidating the molecular mechanisms by which vascular STING activation regulates NK cell function; and resolving the core paradox of being "sensitive yet insufficiently infiltrated."
- Technological Innovation: Developing DynaMITE-seq combined with the 3D MITE model to achieve high-resolution analysis of the dynamic interactions between tumors, blood vessels, and immune cells.
- Therapeutic Innovation: Proposing a combination therapy of STING agonists and DLL3 CAR-NK cells; improving CAR-NK delivery efficiency by activating vascular STING, and providing a translatable strategy for optimizing CAR-NK therapy in solid tumors.
Summary
This study defines the central role of tumor vasculature in SCLC immunosuppression and clarifies that vascular STING activation can break the barrier to immune infiltration, enhancing the killing capacity of CAR-NK cells. These findings fill a gap in the research on the SCLC immune microenvironment, and the combination protocol of STING agonists and CAR-NK cells is expected to bring new therapeutic hope to SCLC patients.
Ubigene consistently upholds the core philosophy of "Making Gene Editing Easier," continuously iterating its products and services. To date, we have achieved over 13,000 successful gene-editing cases and possess 13,000+ cell products (including 10,000+ KO cell lines). Our self-developed innovative technologies have improved gene-editing efficiency by 10-20 times compared to traditional methods. Currently, Ubigene has provided high-quality gene-editing services and products to over 10,000 life science laboratories, pharmaceutical companies, and CROs.
STING is a key signal transduction molecule in the body's innate immune response, triggered by exogenous DNA, and plays a crucial role in regulating the generation of spontaneous anti-tumor immune responses in vivo. If you wish to conduct research on STING, Ubigene can provide STING knockout cell lines, covering popular cell lines such as THP-1, HeLa, and A549. If you have personalized gene-editing needs, please feel free to consult us!
Contact us to learn more>>>


