Targeting LRRC15⁺ CAFs to Suppress Lung Cancer | Research Frontier


Introduction
Lung cancer has the highest global incidence and cancer-related mortality worldwide, with non-small cell lung cancer (NSCLC) accounting for approximately 85% of cases. Although immune checkpoint blockade (ICB) therapy has provided clinical benefits for patients with refractory or metastatic lung cancer, they are only applicable to a small subset of patients, making the development of novel immunotherapeutic strategies urgent. Cancer-associated fibroblasts (CAFs), as core stromal components of the tumor microenvironment (TME), regulate tumor progression and immune evasion through extracellular matrix (ECM) remodeling and growth factor secretion, and exhibit high heterogeneity. Recently, a research team led by Zhu Linnan at Peking University published a study in Cancer Research, identifying LRRC15⁺ CAFs as a tumor-specific CAF subset in lung cancer for the first time. The study demonstrates a key mechanism that LRRC15 promotes ECM production in CAFs, induces M2 macrophage polarization, suppresses CD8⁺ T cell cytotoxicity, and ultimately drives lung cancer progression. Furthermore, the authors developed a bispecific antibody targeting LRRC15 and TGF-β, which effectively downregulates LRRC15 expression in CAFs and inhibits tumor progression, providing a novel therapeutic strategy.

Research Background
CAFs can be classified into subtypes such as myofibroblastic CAFs (myCAFs) and inflammatory CAFs (iCAFs). Among these, LRRC15⁺ CAFs are terminally differentiated myCAFs that have been identified in both pancreatic and lung cancers, where they are associated with tumor progression and ICB resistance. However, the specific molecular mechanisms by which they regulate lung cancer progression remain unclear, and there is a lack of effective therapeutic approaches. LRRC15 belongs to the leucine-rich repeat superfamily and is expressed at low levels in healthy tissues; it may participate in ECM remodeling, but its function within CAFs and its role in regulating the tumor immune microenvironment require elucidation. This study focuses on LRRC15⁺ CAFs, aiming to clarify the potential of LRRC15 as a therapeutic target for lung cancer, elucidate the molecular mechanisms by which it regulates the tumor immune microenvironment, and develop targeted therapeutic strategies.
Research Objectives
To identify CAF subsets and key regulatory molecules with pro-tumorigenic functions in lung cancer, elucidate the molecular mechanisms by which LRRC15 modulates the lung cancer tumor microenvironment and immune responses, validate the efficacy of LRRC15 as a therapeutic target for lung cancer, and develop LRRC15-based targeted therapeutic strategies, thereby providing new directions for lung cancer immunotherapy.
Research Methods
Clinical Samples and Database Analysis
The study included clinical samples from 14 pathologically confirmed lung cancer patients. It integrated 12 single-cell RNA sequencing (scRNA-seq) datasets related to lung adenocarcinoma (11 public datasets + 1 in-house dataset), along with public databases such as TCGA, GTEx, and GEPIA, to analyze the distribution of LRRC15⁺ CAFs and the correlation between LRRC15 expression and the prognosis of lung cancer patients.
Animal Models
Genetically engineered mouse models, including C57BL/6J, Lrrc15 knockout, fibroblast-specific Lrrc15 conditional knockout (Col1a2-CreER; Lrrc15fl/fl), and M-NSG mice were used. Subcutaneous tumor models (TC1-GFP, LLC, KPS) and a LLC-Luc lung metastasis model were established to evaluate the impact of LRRC15 deficiency and targeted antibody therapy on tumor growth. Macrophages were depleted using clodronate liposomes to validate their role in LRRC15-regulated tumor progression.
Cellular Experiments
Mouse lung fibroblasts and human patient-derived lung cancer fibroblasts were induced to differentiate into LRRC15⁺ CAFs using TGF-β. CRISPR-Cas9 technology was used to generate human/mouse LRRC15-KO CAF cell lines. Mouse bone marrow-derived macrophages (BMDMs), human peripheral blood monocyte-derived macrophages (mo-MΦs), mouse splenic CD8⁺ T cells, and human peripheral blood CD8⁺ T cells were isolated. Co-culture, Transwell, and ECM co-culture experiments were performed to validate the regulatory effects of LRRC15 on macrophage polarization and CD8⁺ T cell activation.
Molecular Mechanism Validation
scRNA-seq, bulk RNA-seq, qPCR, and Western blot were used to detect gene and protein expression. Multiplex immunohistochemistry (mIHC) and flow cytometry were used to analyze immune cell infiltration and phenotypes within tumor tissues. Sirius Red staining and collagen concentration assays were used to assess ECM (collagen) production. Immunofluorescence was used to validate the in vivo targeting of the bispecific antibody.
Therapeutic Validation
A bispecific antibody targeting LRRC15 and TGF-β was designed and generated. Its efficacy as a monotherapy was evaluated in a KPS lung cancer mouse model, assessing its effects on tumor growth, LRRC15 expression, ECM deposition, and the immune microenvironment.
Research Strategy
- Identification of Lung Cancer CAF Subsets: Construction of a human lung stromal cell scRNA-seq atlas to identify tumor-enriched CAF subsets associated with poor prognosis and screen for their specific surface molecules;
- Functional Validation of LRRC15: Demonstration in cellular and animal models that LRRC15 deficiency suppresses lung cancer progression in a host immune system-dependent manner;
- Elucidation of Immune Regulation Mechanism: Clarification of the molecular pathway by which LRRC15 promotes ECM production in CAFs, induces CD206⁺ M2 macrophage polarization, and subsequently inhibits CD8⁺ T cell infiltration and cytotoxic function;
- Development of Targeted Therapeutic Strategy: Design and validation of a bispecific antibody targeting LRRC15 and TGF-β, demonstrating its therapeutic efficacy and safety in lung cancer models.
Key Findings
1. LRRC15⁺ CAFs are Enriched in Lung Cancer Tissues and Correlate with Poor Patient Prognosis
By integrating 12 lung stromal scRNA-seq datasets, a stromal atlas comprising 26,091 high-quality cells was constructed, identifying nine fibroblast subclusters. Among them, LRRC15⁺ CAFs (c03) were significantly enriched in lung cancer tissues, accounting for approximately 40% of fibroblasts in tumor samples, but were rarely detected in adjacent normal tissues. mIHC results showed abundant LRRC15⁺α-SMA⁺ CAFs within tumor nests of lung cancer patients, with almost no expression in adjacent normal tissues. Scissor analysis identified LRRC15⁺ CAFs as the fibroblast subset most significantly associated with poor prognosis in lung cancer. TCGA data further demonstrated that high expression of LRRC15⁺ CAF signature genes correlates with reduced overall survival in lung, colorectal, and gastric cancers, high LRRC15 gene expression was significantly associated with poor prognosis in lung cancer patients. Furthermore, LRRC15 showed low expression in healthy tissues and was specifically expressed in lung cancer CAFs, with minimal expression in immune cells, epithelial cells, or endothelial cells, suggesting its potential as a therapeutic target with good specificity and safety.
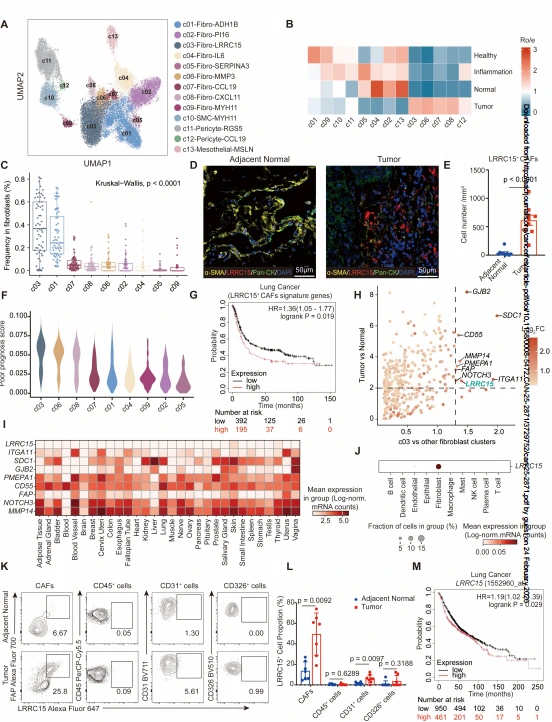
Figure 1. LRRC15+ stromal cells are enriched in lung tumors and correlate with poor patient prognosis.
2. LRRC15 Deficiency Suppresses Lung Cancer Progression in An Immune-Dependent Manner
TGF-β was shown to induce mouse lung fibroblasts to upregulate LRRC15 expression and differentiate into LRRC15⁺ CAF-like cells. Co-injection of TGF-β-induced WT or Lrrc15-KO CAFs with lung cancer cells into C57BL/6J mice showed that Lrrc15-KO CAFs significantly suppressed the growth of TC1-GFP and LLC subcutaneous tumors, an effect further enhanced by combined bleomycin and TGF-β treatment. In Lrrc15 knockout and fibroblast-specific conditional knockout mice, KPS tumor growth was markedly suppressed, lung metastasis (LLC-Luc) was reduced, and survival was prolonged. However, in immunodeficient NSG mice, there was no difference in tumor growth between WT and Lrrc15-KO CAF co-injection groups. Moreover, in vitro co-culture experiments showed that LRRC15 deficiency did not affect the direct impact of CAFs on lung cancer cell proliferation, indicating that the anti-tumor effect of LRRC15 deficiency depends on the host immune system.
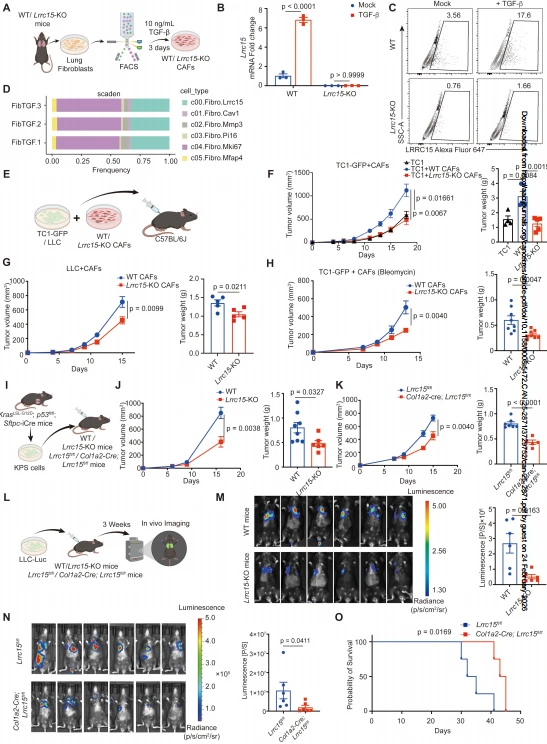
Figure 2. LRRC15 deficiency in CAFs suppresses tumor progression.
3. LRRC15 Deficiency Remodels the Tumor Immune Microenvironment by Enhancing CD8⁺T Cell Activation and Inhibiting M2 Macrophage Polarization
Flow cytometry analysis revealed that tumors co-injected with Lrrc15-KO CAFs showed increased infiltration of CD45⁺ immune cells and lymphocytes. Specifically, the proportion of CD8⁺ T cells was significantly elevated, along with upregulated expression of perforin, granzyme B, and IFN-γ, indicating enhanced cytotoxic function. Within the myeloid cell compartment, the proportion of infiltrating macrophages decreased, and the ratio of CD206⁺ M2 macrophages to CD206⁻ macrophages was significantly reduced. mIHC validation confirmed increased CD8⁺ T cell infiltration and decreased F4/80⁺ macrophage infiltration in tumors from the Lrrc15-KO CAF group. TCGA analysis further showed that LRRC15⁺ CAF signature gene expression positively correlates with M2 macrophage markers, and that the density of LRRC15⁺ CAFs in tumor tissues was significantly positively correlated with CD68⁺ macrophage density, suggesting that LRRC15 regulates the functional balance between macrophages and CD8⁺ T cells in lung cancer.
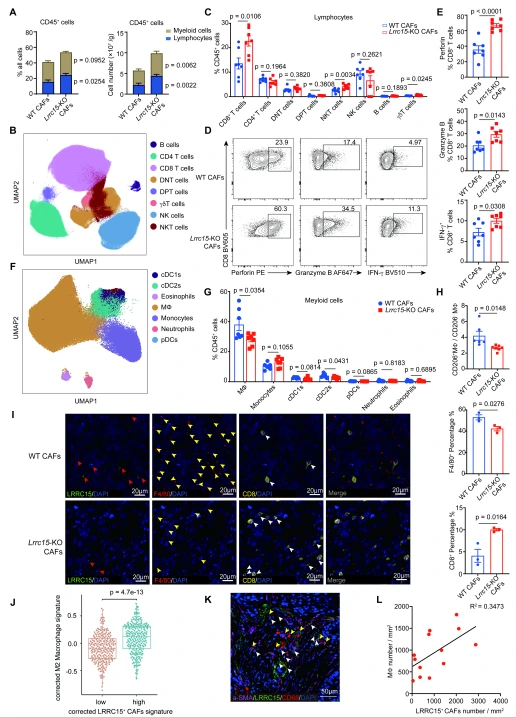
Figure 3. LRRC15 deficiency in CAFs induces increased CD8⁺ T cell abundance and activation, and reduces macrophage infiltration.
4. LRRC15 in CAFs Drives M2 Macrophage Polarization and Indirectly Suppresses CD8⁺T Cell Activation
In vitro co-culture experiments demonstrated that wild-type CAFs significantly increased the proportion of CD206⁺ macrophages in both mouse BMDMs and human monocyte-derived macrophages (mo-MΦs), whereas Lrrc15/LRRC15 knockout CAFs almost completely abolished this effect. Tumor-derived Lrrc15-KO LRRC15⁺ CAFs also markedly inhibited M2 polarization. Macrophage depletion experiments using clodronate liposomes completely reversed the tumor-suppressive effect observed with Lrrc15 conditional knockout, confirming that macrophages are a critical mediator of LRRC15-regulated lung cancer progression. Furthermore, while direct co-culture of CAFs with CD8⁺ T cells showed that LRRC15 deficiency did not directly affect CD8⁺ T cell activation, macrophages pre-cultured with Lrrc15-KO CAFs significantly promoted CD8⁺ T cell proliferation and expression of granzyme B and IFN-γ, indicating that LRRC15 indirectly suppresses CD8⁺ T cell function by regulating macrophage polarization.
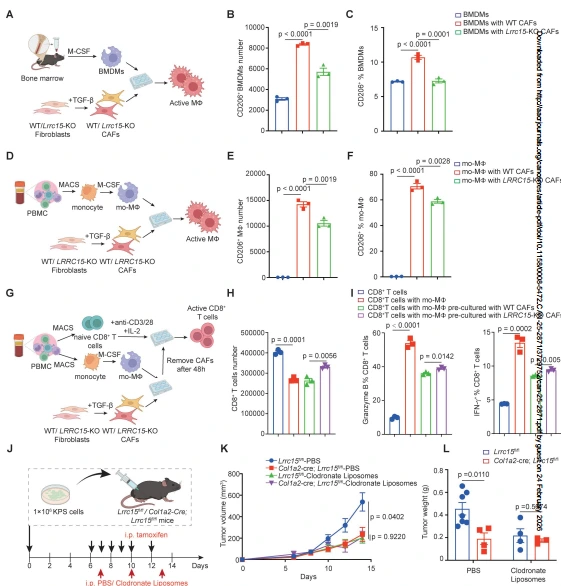
Figure 4. LRRC15 in fibroblasts drives M2 macrophage polarization to suppress CD8⁺ T cell activation.
5. LRRC15 Regulates M2 Macrophage Polarization by Promoting ECM Production in CAFs
Bulk RNA-seq and GO analysis revealed that ECM-related pathways were significantly enriched in WT CAFs compared to LRRC15-KO CAFs. LRRC15 expression positively correlated with ECM-related gene expression in lung cancer patients, and ECM organization pathways were enriched in LRRC15⁺ CAFs in both humans and mice. Sirius Red staining, collagen quantification, and Western blot analysis demonstrated that LRRC15 deficiency significantly reduced collagen (COL1A1) production in both human and mouse CAFs, and ECM density is significantly decreased in tumors from Lrrc15 conditional knockout mice. When macrophages were cultured on decellularized ECM derived from CAFs, the ECM from Lrrc15/LRRC15-KO CAFs significantly reduced the number and proportion of CD206⁺ macrophages. Transwell experiments and cytokine neutralization assays confirmed that LRRC15-regulated macrophage polarization was independent of soluble cytokines but dependent on direct contact with CAFs. Exogenous collagen I supplementation significantly increased CD206⁺ macrophage proportions, confirming that LRRC15 promotes M2 polarization by enhancing ECM (collagen I) production.
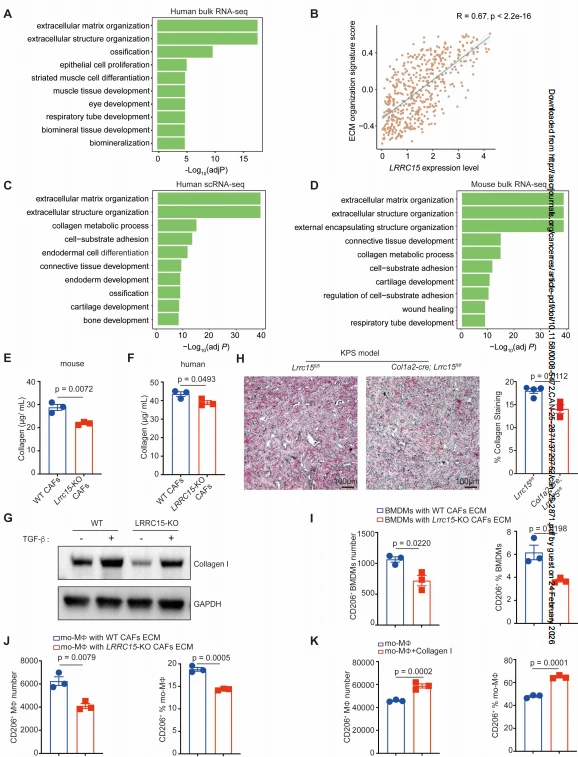
Figure 5. LRRC15 promotes M2 polarization by modulating the extracellular matrix production.
6. Bispecific Antibody Targeting LRRC15 and TGF-β Effectively Suppresses Lung Cancer Progression with Improved Safety
The generated anti-LRRC15 antibody (Abio-1) shows high affinity for both human and mouse LRRC15. Based on the role of TGF-β in inducing LRRC15⁺ CAF differentiation, a bispecific antibody targeting LRRC15 and TGF-β was developed. In vivo tracing showed that this antibody specifically accumulated in LRRC15⁺ CAFs within tumor tissues, with minimal binding to immune cells or tumor cells. In the KPS lung cancer model, the bispecific antibody significantly inhibited tumor growth, achieving efficacy comparable to that of a TGF-βRII antibody, while the anti-LRRC15 antibody alone showed no significant anti-tumor effect. Unlike the TGF-βRII antibody, which caused splenomegaly (a side effect of systemic TGF-β blockade), the bispecific antibody did not induce this phenotype, suggesting it can block TGF-β signaling locally by targeting LRRC15, thereby offering improved safety. Additionally, bispecific antibody treatment significantly reduced LRRC15 expression levels in LRRC15⁺ CAFs (without eliminating the CAFs) and decreased tumor ECM deposition. In immunodeficient NSG mice, the bispecific antibody showed no anti-tumor effect, confirming that its efficacy depends on the host immune system.
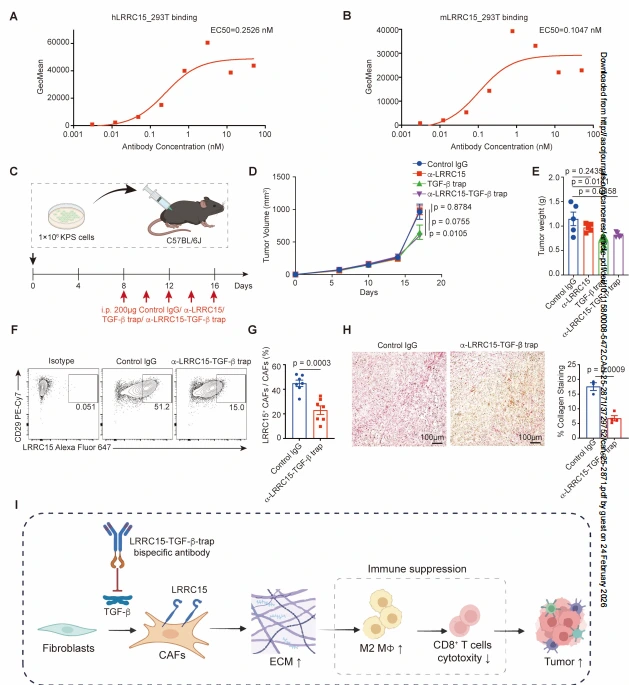
Figure 6. Targeting LRRC15-positive cancer-associated fibroblasts inhibits tumor progression.
Significance and Novelty
Mechanistic Innovation
This study systematically elucidates the molecular mechanism by which LRRC15⁺ CAFs regulate the lung cancer tumor immune microenvironment for the first time. It demonstrates that LRRC15 promotes ECM production in CAFs, thereby inducing M2 macrophage polarization and subsequently suppressing CD8⁺ T cell infiltration and cytotoxic function. These findings establish a regulatory axis of CAFs–ECM–macrophages–CD8⁺ T cells as a central mechanism driving lung cancer progression, thereby expanding current understanding of CAF heterogeneity and functional regulation.
Target Identification
This study provides the first evidence that LRRC15 is a functional driver molecule in tumor-specific CAFs in lung cancer. Its low expression in normal tissues and specific expression in LRRC15⁺ CAFs highlight its suitability as a therapeutic target, offering a novel molecular target for precision treatment of lung cancer.
Therapeutic Strategy
The study develops a bispecific antibody targeting LRRC15 and TGF-β, which selectively targets LRRC15⁺ CAFs within tumor tissues, downregulates LRRC15 expression, and reduces ECM deposition, thereby effectively inhibiting lung cancer progression while avoiding the side effects associated with systemic TGF-β blockade. This provides a novel and feasible strategy for CAF-targeted immunotherapy in lung cancer and offers a reference for CAF-targeted therapy in other solid tumors.
Summary
By constructing a human lung stromal cell atlas based on scRNA-seq, this study identifies LRRC15⁺ CAFs as a tumor-enriched CAF subset associated with poor prognosis in lung cancer and establishes LRRC15 as the key functional molecule of this subset . This study not only reveals a novel mechanism by which CAFs regulate lung cancer immunity but also identifies a highly promising therapeutic target for lung cancer and develops a corresponding targeted therapeutic strategy, providing important theoretical and experimental foundations for overcoming immunotherapy resistance and improving treatment outcomes in lung cancer.
Ubigene Genome Editing Services
Ubigene has always adhered to its core mission of "Make genome editing easier," continuously iterating its products and services. To date, Ubigene has successfully completed over 13,000 gene editing projects and offers a comprehensive portfolio of more than 11,000 cell products, including over 8,000 knockout (KO) cell lines. Leveraging proprietary technologies, Ubigene achieves 10–20 fold higher gene editing efficiency compared to traditional methods. Ubigene has provided high-quality gene editing services and products to over 10,000 life science laboratories, pharmaceutical companies, and CROs worldwide.
LRRC15 is a type I transmembrane protein involved in cell-cell and cell-matrix interactions and has been found to be closely associated with the proliferation, migration, and invasion of tumor cells. If you are also interested in conducting research on LRRC15, Ubigene can provide LRRC15 knockout cell lines, covering tumor cell lines such as U-87 MG and Ishikawa . If you have customized gene editing needs, Contact us to learn more>>>
Reference
Qi L, Dang G, Ling X, Miao Y, Bo Y, Zhai Y, Chen X, Zhai Q, Zheng L, Zhang Y, Li Y, Liu C, Fan H, Si W, Tong D, Yang Z, Hu X, Wang D, Cheng S, Zhang Z, Zhu L. Targeting LRRC15 in Cancer-Associated Fibroblasts Modifies the Extracellular Matrix and Enhances Tumor Immune Responses to Suppress Lung Cancer Progression. Cancer Res. 2026 Jan 27. doi: 10.1158/0008-5472.CAN-25-2871. Epub ahead of print. PMID: 41591362.



