Expert Insights | Practical Tips for LLC Cell Culture and Gene Editing


LLC cells are derived from the lungs of C57BL mice and are widely used as a metastasis model for studying the mechanisms of cancer chemotherapy drugs. These cells are reported to be highly metastatic and drug-resistant mouse tumor cells. This document provides exclusive culture techniques for LLC cells, offering a comprehensive guide to mastering LLC cell culture and key gene editing practices.
Overview of Mouse Lung Cancer Cells (LLC)
- Cell Name: Mouse Lung Cancer Cells (LLC)
- Cell Morphology: Mixed population of epithelial-like and round cells
- Culture Medium: 90% DMEM+10% FBS
- Atmosphere: Air, 95%; Carbon dioxide, 5%
- Temperature: 37℃
- Medium Renewal Frequency: Remove when passaging
- Passage Ratio: 1:2 - 1:3
Reference for Cell Growth Status:
Normal Morphology: Heterogeneous cell morphology, with both epithelial-like and round cells present in varying proportions; both types adhere to the culture surface. A small number of suspended cells may be observed.
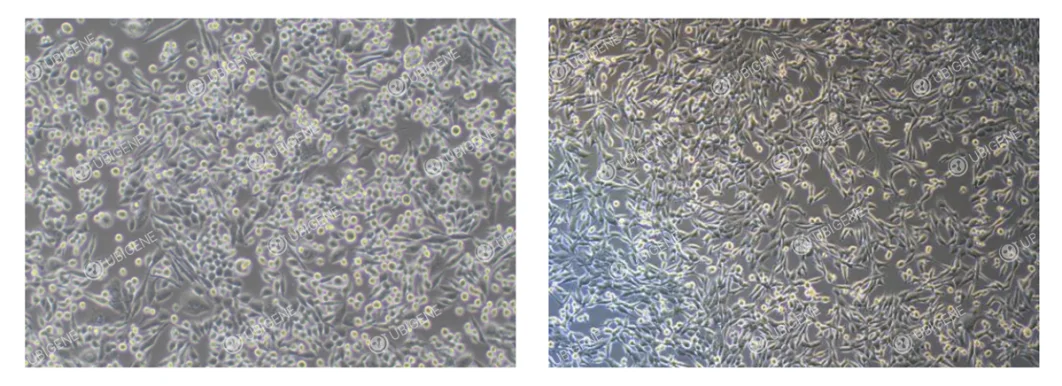
Abnormal Morphology: Cells appear spindle-shaped or elongated; cell shrinkage or rounding; increased cell debris.
LLC Cell Thawing Procedure
- Prepare Culture Medium Add 7 mL of complete culture medium to a centrifuge tube and keep it ready.
- Thaw Cells Remove the vial from dry ice. Using forceps, hold the vial cap and immerse it in a 37°C water bath. Shake gently (ensure the cap remains above the water surface). Thaw quickly for approximately 1 minute until only a small ice crystal remains. Stop water bath.
- Centrifuge Cells Transfer the thawed cell suspension to a centrifuge tube. Centrifuge at 1100 rpm for 4 minutes and discard the supernatant.
- Resuspend and Seed Cells Resuspend the cell pellet in complete culture medium and seed into an appropriately sized culture dish or flask.
- Culture Cells Place the culture dish or flask in a 37°C incubator. After 24 hours, observe cell attachment and morphology.
LLC Cell Subculture Procedure (using a T25 flask as an example)
-
Subculture Conditions
Subculture cells when they reach 80-90% confluency.
In a biosafety cabinet, discard the culture medium and wash the cells 1-2 times with 5 mL PBS. -
Trypsin Digestion
Add 1 mL of trypsin, gently swirl the flask to ensure the enzyme fully covers the cells.
Place the flask in a 37°C incubator for 1-2 minutes.
Under a microscope, when most cells become round and bright, gently tap the sides of the flask to detach cells, and immediately stop the digestion. -
Terminate Digestion
Add 2 mL of complete culture medium (2* the volume of trypsin) to stop the reaction.
Transfer the cell suspension to a 15 mL centrifuge tube. -
Centrifuge Cell Suspension
Centrifuge at 1100 rpm at room temperature for 4 minutes.
Discard the supernatant and resuspend the cell pellet in complete culture medium. 离心结束,弃去上清,加入完全培养基重悬细胞。 -
Subculture and Culture
Seed cells at a split ratio of 1:2-1:3.
Observe cell morphology and attachment the next day.
LLC Cell Cryopreservation Procedure
-
Collect Cells
Harvest trypsinized cells following the standard subculture procedure and transfer them into a centrifuge tube. -
Centrifugation
Centrifuge at 1100 rpm for 4 minutes and discard the supernatant. -
Resuspend and Aliquot for Freezing
Resuspend the cell pellet in cryopreservation medium. Adjust the concentration to 1*10^6 cells/mL and aliquot 1 mL per cryovial. Label each vial with cell line name, passage number, and date. -
Cooling and Storage
Place the vials in a controlled-rate freezing container and store overnight at -80°C. Transfer the vials into liquid nitrogen for long-term storage.
LLC Cell Culture Notes
-
Cell Confluency: Subculture cells at 80-90% confluency for optimal growth.
-
Medium Storage: Store culture medium at 4°C protected from light and use within the expiration date.
-
Culture Environment: Maintain a stable and appropriate incubator environment.
-
Operational Details: Pre-warm culture medium and trypsin to 37°C before use to avoid temperature stress.
-
Subculture Handling: Avoid excessive pipetting to prevent cell membrane damage; gently swirl the flask if cells are unevenly attached to achieve uniform distribution.
-
Use High-Quality Serum: LLC cells are sensitive to serum quality. High-quality fetal bovine serum (FBS) is required to maintain adherent growth; poor serum quality may alter cell morphology and behavior.
Tip: A small number of suspended cells may be present during culture. If adherent cells constitute the majority, suspended cells can be discarded. If many suspended cells are present, they can be collected by centrifugation and re-seeded into the culture flask.
Precautions for LLC Cell Transfection
-
Cell Condition Requirements
- Ensure cells are healthy and in the logarithmic growth phase, with 70-80% confluency.
- Cell viability should be >80%, which can be assessed using Trypan Blue exclusion.
- Use low-passage cells.
- Carefully monitor trypsinization time to avoid over-digestion and cell damage.
- During the procedure, generate a single-cell suspension and avoid cell clumping.
-
Transfection Reagents and Pre-Experiment
- Mix transfection reagents thoroughly before use to ensure uniformity.
- Perform preliminary antibiotics-selection experiments to determine the optimal selection concentration post-transfection.
-
Electroporation
- Control the number of cells and seed them into appropriately sized culture plates after electroporation.
- Use gentle trypsin and terminate digestion completely with serum-containing medium.
- Wash cells 1-2 times with PBS to completely remove residual serum, preventing ionic interference with electroporation.
- Optimize electroporation parameters through preliminary experiments.
- Ensure post-electroporation cell attachment rate is ≥50%.
- Keep total electroporation time short to avoid excessive stress.
-
Lentiviral Transduction
- Perform preliminary experiments to determine the optimal MOI.
- Maintain cell confluency at 30-40% before infection; higher confluency should be avoided.
- Add Polybrene prior to infection to enhance viral uptake.
- Change medium 24 hours after infection.
- Avoid repeated freeze-thaw cycles of viral stocks.
- If infection efficiency is low, consider repeat infection (ensuring cell tolerance) or use centrifugation-enhanced transduction.
Precautions for ARPE-19 Single-cell cloning experiment
-
Cell Condition Requirements
- Use cells in the logarithmic growth phase for single-clone seeding. Before seeding, maintain approximately 60-70% confluency.
- Cell viability should be ≥80% at the time of seeding.
-
Reagents and Pre-Experiment Preparation
- Pre-warm all reagents, including culture medium, trypsin, and PBS, prior to the experiment.
- Use gentle dissociation reagents, such as TrypLE Express, to minimize cell damage.
-
Seeding Strategy
- Conduct preliminary experiments to determine the optimal seeding density gradient, avoiding too low a single-clone formation rate.
- When seeding 96-well plates, ensure even cell distribution. Fill the outer wells with PBS to prevent evaporation.
- Avoid frequently moving the plate during clone culture to minimize the risk of single-cell clone detachment or migration.
-
Dilution Method
- Employ the limiting dilution method for single-clone seeding.
- After cell counting and dilution, the optimal cell number should fall within 1 * 10⁶ to 2 * 10⁶ cells.
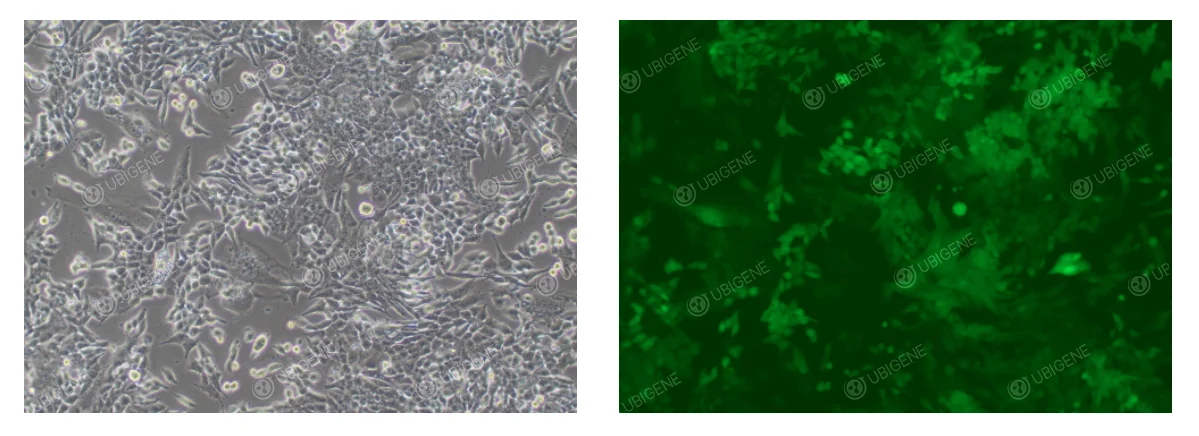
Cell Images After lentiviral infection
Common Issues and Solutions in LLC Cell Culture
How to Address Pathological Cell Clumping?
Observation: Large, dense cell clusters visible under the microscope; light difficult to penetrate the clumps; the center of the clumps appears dark (indicating potential hypoxia or necrosis).
-
Possible Causes:
- Excessive cell density.
- Improper trypsinization.
- Issues with culture medium or serum.
-
Solutions:
- Subculture promptly when cell confluence reaches 80%.
- Subculture promptly when cell confluence reaches 80%.
- When resuspending cells, gently pipette multiple times to achieve a single-cell suspension.
- Use high-quality fetal bovine serum for culture.
How to Address Slow or Stalled Cell Growth?
Observation: Slow proliferation after subculture; prolonged time to reach confluent density; slow or no change in medium color.
-
Possible Causes:
- Cell density too low.
- Nutrient depletion in medium (e.g., glutamine deficiency).
- Mycoplasma contamination.
- Cell senescence (excessively high passage number).
-
Solutions:
- Increase seeding density: Use a ratio of 1:2 for the first few passages; avoid seeding too sparsely.
- Use fresh medium: Ensure freshly prepared complete medium is used (stored at 2-8°C, ideally used within one week).
- Check for contamination: If operational errors are excluded, perform mycoplasma testing.
- Use low-passage-number cells.
Ubigene LLC Cell lineRelated products
Ubigene provides stable and reliable LLC cells. Welcome to search our Red Cotton OmniCell Bank. All human and mouse cell lines are provided with STR authentication reports to ensure cell line identity accuracy. Ubigene offers over 1,000 wild-type cell lines covering multiple research fields, along with specialized cell culture media, to comprehensively support your scientific research needs.
LLC cells are widely used as a metastasis model for studying the mechanisms of cancer chemotherapy drugs. Leveraging advanced gene editing technology and platform, Ubigene has developed gene knockout, Luciferase, EGFP, and Cas9 stable cell lines based on this cell line. We also offer gene modification services for LLC cells, including gene knockout, point mutation, gene knock-in, overexpression, and interference. For inquiries, please contact us.
Contact us to learn more>>>


