Expert Insights | Practical Tips for HTR-8/SVneo Cell Culture and Gene Editing


HTR-8/SVneo cells were derived from first-trimester placental villous tissue (approximately 6–8 weeks of gestation) obtained from a spontaneous abortion. Human first-trimester placental villous explants were transfected with a gene encoding the Simian Virus 40 (SV40) large T antigen, resulting in the establishment of an immortalized human extravillous trophoblast (EVT) cell line, HTR-8/SVneo. These cells express multiple markers characteristic of trophoblasts, including: Insulin-like growth factor II (IGF-II), NDOG-5, Proliferating cell nuclear antigen (PCNA), Human leukocyte antigen class I framework antigen (W6/32), and a distinct set of integrins.
HTR-8/SVneo cells are widely used for functional studies of trophoblasts and placental biology. Notably, these cells are negative for the macrophage marker 63/D3, the endothelial marker von Willebrand factor (factor VIII), and the integrin subunits α6 and β4, supporting their trophoblast-specific identity. Here, we present exclusive HTR-8/SVneo culture protocols, providing researchers with practical guidance for efficient cell maintenance as well as key considerations for gene editing applications, unlocking the full potential of this widely used human extravillous trophoblast model.
Basic Information on Human Trophoblast Cell Line (HTR-8/SVneo)
- Cell Name: Human Extravillous Trophoblast Cells (HTR-8/SVneo)
- Cell Morphology: Epithelial-like cells, adherent growth
- Culture Conditions: 90% DMEM + 10% Fetal Bovine Serum (FBS)
- Atmosphere: 95% air, 5% CO₂
- Temperature: 37°C
- Medium Change Frequency: Every 2–3 days
- Subculture Ratio: 1:2 to 1:4
Cell Growth Characteristics:
Normal Condition: Cells exhibit a polygonal or irregular shape, closely apposed to one another, forming a confluent monolayer that resembles a cobblestone pattern. Cells are organized with clear boundaries. At low density, cell bodies may appear slightly elongated, with occasional spindle-shaped or triangular cells, displaying a fibroblast-like morphology. (As shown below)
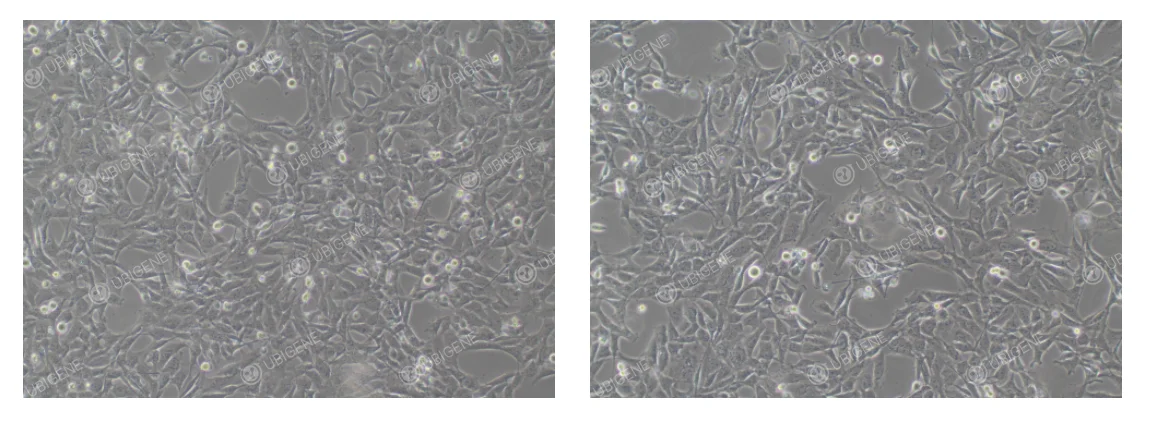
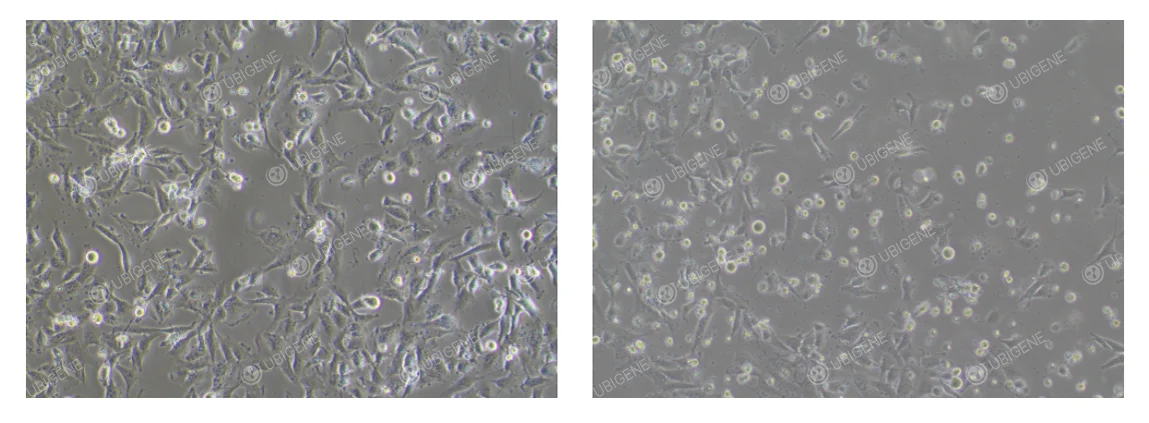
Abnormal Condition: Cells appear stretched or elongated and exhibit irregular shapes. They may become atrophied or rounded, with weak adherence to the culture surface. Cell debris may increase, and transparent cytoplasmic vacuoles can be observed. (As shown below)
HTR-8/SVneo Cell Thawing and Subculture Protocols
-
Prepare Complete Medium
Aliquot 7 mL of complete culture medium into a sterile centrifuge tube and keep ready. -
Cell Thawing
Remove the vial from dry ice and hold the cap with sterile forceps. Place the vial in a 37°C water bath, gently swirling to accelerate thawing. Ensure water does not cover the cap. Thaw for approximately 1 minute, until ice crystals are reduced to pea-sized fragments, then remove the vial from the water bath. -
Centrifugation
Transfer the thawed cell suspension to a centrifuge tube. Centrifuge at 1,100 rpm for 4 minutes at room temperature. Carefully discard the supernatant. -
Resuspension and Seeding
Resuspend the cell pellet in complete culture medium. Seed cells into appropriately sized culture dishes or flasks. -
Cell Culture
Place the culture dish or flask in a 37°C incubator. After 24 hours, observe cell attachment and morphology.
Subculture Procedure (Example: T25 Flask)
-
Subculture Criteria
Passage cells when confluency reaches 80–90%.
In a biosafety cabinet, remove the culture medium and wash the cells 1–2 times with 5 mL PBS. -
Trypsinization
Add 1 mL of trypsin, gently swirling to ensure all cells are covered.
Incubate the flask in a 37°C incubator for 2–3 minutes.
Under a microscope, observe for cells becoming round and bright. Gently tap the sides of the flask; when most cells detach, immediately stop the digestion. -
Termination of Digestion
Add 2 mL of complete culture medium (twice the trypsin volume) to neutralize trypsin.
Transfer the cell suspension to a 15 mL centrifuge tube. -
Cell Centrifugation
Centrifuge at 1,100 rpm for 4 minutes at room temperature.
Discard the supernatant and resuspend the cell pellet in complete medium. -
Cell Seeding for Subculture
Passage cells at a 1:2 to 1:4 split ratio.
Observe cell morphology and attachment the following day.
HTR-8/SVneo Cell Cryopreservation Protocol
-
Cell Collection
Harvest cells as per the subculture procedure and transfer the trypsinized cells into a centrifuge tube. -
Centrifugation
Centrifuge at 1,100 rpm for 4 minutes and discard the supernatant. -
Resuspension and Freezing
Resuspend the cell pellet in cell freezing medium. Aliquot 1 × 10⁶ cells per 1 mL freezing medium into cryovials. Label vials with cell name, passage number, and date. -
Controlled Cooling and Storage
Place cryovials in a programmable freezing container and store at -80°C overnight. Transfer vials to liquid nitrogen for long-term storage.
HTR-8/SVneo Cell Culture Considerations
- Cell Confluency: Passage cells when they reach 80–90% confluency.
- Medium Replacement: Replace complete culture medium every 2–3 days for actively growing cells.
- Medium Storage: Store medium at 4°C, protected from light, and use before expiration.
- Culture Environment: Maintain optimal incubation conditions (37°C, 5% CO₂, humidified).
- Operational Details: Pre-warm medium and trypsin to 37°C before use to avoid temperature stress.
- Subculture Tips: Avoid excessive pipetting to prevent cell membrane damage. If cells are unevenly attached, gently shake the flask to distribute cells evenly.
HTR-8/SVneo Cell Transfection Guidelines
-
Cell Condition Requirements
- Use healthy cells in the logarithmic growth phase (70–80% confluency).
- Cell viability should be >80%, as determined by trypan blue exclusion.
- Prefer low-passage cells.
- Avoid over-trypsinization to prevent cell damage.
- Ensure a single-cell suspension to prevent clumping during handling.
-
Transfection Reagent and Pre-testing
- Mix transfection reagents thoroughly to ensure uniformity.
- Conduct preliminary drug selection tests to determine optimal concentrations for post-transfection selection.
-
Electroporation Method
- Control cell number and seed cells into appropriate plates post-electroporation.
- Use mild trypsin and neutralize with serum-containing medium.
- Wash cells 1–2 times with PBS to remove residual serum and avoid ionic interference.
- Optimize electroporation parameters via pre-experiments.
- Ensure cell attachment ≥60% after electroporation.
- Keep total electroporation time brief to minimize cell stress.
-
Lentiviral Transduction
- Perform pre-testing to determine the optimal MOI.
- Seed cells at 30–40% confluency prior to infection; avoid high confluency.
- Add Polybrene to enhance infection efficiency.
- Replace medium 24 hours post-infection.
- Avoid repeated freeze-thaw cycles of viral stocks.
- If infection efficiency is low, consider re-infection (ensuring cell tolerance) or spinoculation methods.
HTR-8/SVneo Single-Cell Cloning Guidelines
-
Cell Condition Requirements
- Use log-phase cells for clonal seeding. Prior to seeding, confluency should be approximately 70%.
- Cell viability should be ≥80% at the time of seeding.
-
Reagents and Pre-testing
- Pre-warm all reagents, including culture medium and PBS, before use.
- It is recommended to use gentle dissociation reagents (e.g., TrypLE Express) to minimize cell stress.
-
Cell Seeding Strategy
- Perform preliminary experiments to determine the optimal seeding density, ensuring sufficient single clones while avoiding overly sparse distribution.
- When seeding into 96-well plates, ensure even cell distribution. Add PBS to the outer wells to prevent evaporation.
-
Limiting Dilution Method
- Use the limiting dilution method for single-cell seeding.
- After calculating the dilution, the final cell number per well should ideally support proper clonal formation. (补写:Typical target range is 0.5–2 cells per well statistically.)
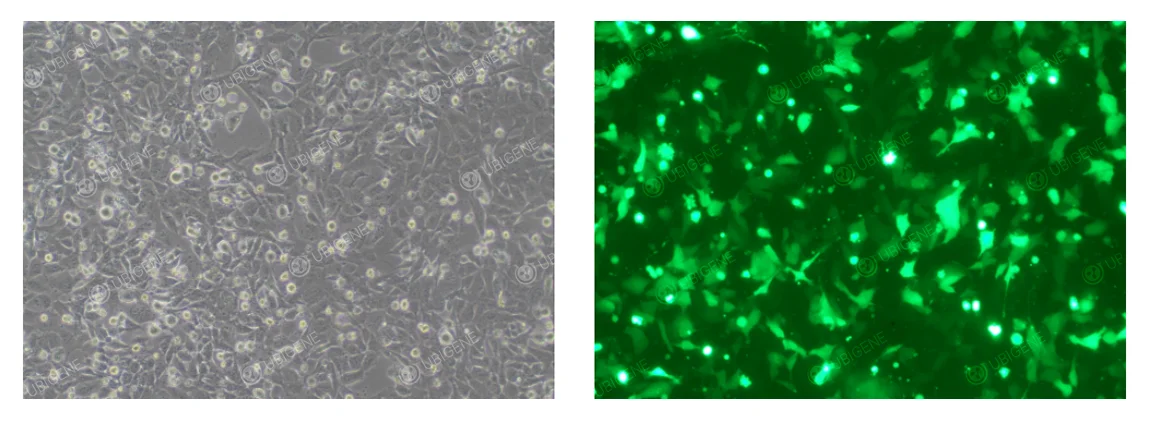
HTR-8/SVneo Cells After Electroporation
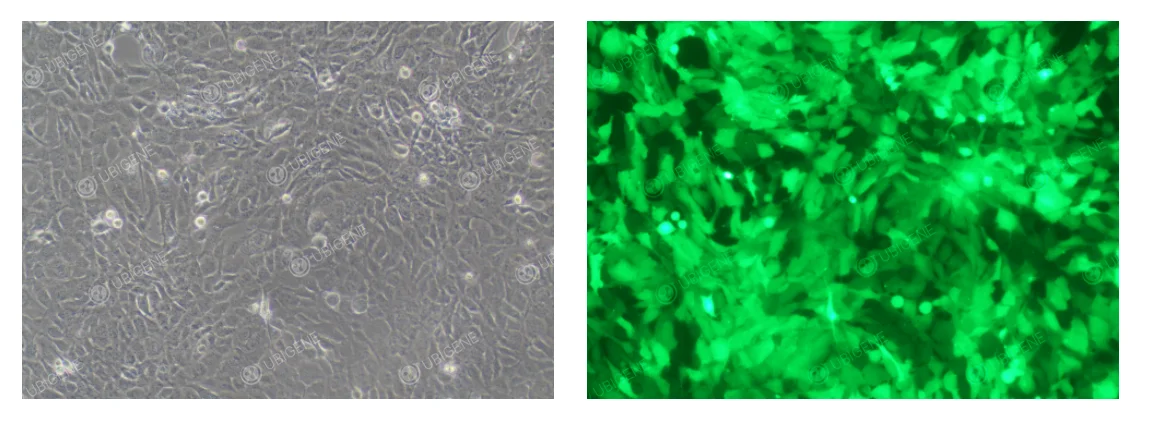
HTR-8/SVneo Cells After Lentiviral Infection
Common Issues and Troubleshooting in HTR-8/SVneo Cell Culture
1. Severe Cell Detachment
Observation: Under the microscope, small, bright, round cells are floating in the medium. In severe cases, large sheets of cells (still partially connected) may lift from the culture surface, curl, and float in the medium.
Possible Causes:
- Handling is too vigorous (e.g., pipetting or shaking).
- Over-digestion with trypsin.
- Passaging cells at excessive confluency.
- Trypsin activity is too strong or used in excess.
Solutions:
- Handle cells very gently during all culture operations.
- Strictly control trypsin digestion time, checking under the microscope at 1-minute intervals.
- Passage cells at 80–90% confluency, avoiding 100% confluency.
- Use standard trypsin concentration, or try lower concentration (e.g., 0.05%) with extended digestion time if necessary.
2. Abnormal Cell Morphology
Observation: Cells appear excessively elongated; increased granularity and vacuolation are visible.
Possible Causes:
- Widespread granules or vacuoles: may be due to serum toxicity or accumulation of metabolic waste.
- Excessive cell elongation and loss of epithelial-like morphology: may result from over-digestion with trypsin or too low seeding density.
Solutions:
- Use high-quality, validated fetal bovine serum (FBS); avoid serum of unknown origin. Increase medium replacement frequency.
- Optimize trypsin digestion time (generally 2–3 minutes); adjust seeding density appropriately.
- Small-scale vacuolation in a few cells is usually natural aging or apoptosis and is considered normal.
Ubigene HTR-8/SVneo Cell-Related Products
Ubigene provides stable and reliable HTR-8/SVneo cells for your research. You could explore our Red Cotton OmniCell Bank, which includes a wide range of human and mouse cell lines. All cells come with STR authentication reports to ensure accurate cell identity. Ubigene offers over 1,000 wild-type cell lines covering multiple research areas, and also provides cell-type specific culture media to fully support your research needs.
HTR-8/SVneo cells are widely used in studies of trophoblast function and placental biology. Leveraging our mature gene-editing technologies and platforms, Ubigene has generated HTR-8/SVneo cell lines with gene knockouts as well as stable Luc, EGFP, and Cas9-expressing lines. We also provide a full range of gene modification services for this cell line, including gene knockout, point mutation, gene knock-in, overexpression, and gene knockdown/interference.
Contact us for more information >>>


