Research Frontiers | Deciphering a Core Mechanism: The m⁶A Reader YTHDF1 Drives Colorectal Cancer Stemness and Chemoresistance by Regulating NOTCH1 Translation


Introduction
Colorectal cancer (CRC) is the third most commonly diagnosed malignancy and the second leading cause of cancer-related mortality worldwide. Chemoresistance and tumor recurrence remain major obstacles to improving patient outcomes, with cancer stem cells (CSCs) widely recognized as the central drivers of both chemoresistance and relapse. N⁶-methyladenosine (m⁶A) modification, the most prevalent epitranscriptomic regulation in eukaryotic mRNA, plays a pivotal role in tumor initiation and progression. In a recent study published in Signal Transduction and Targeted Therapy, research teams led by Jun Yu and Zijun Huang at The Chinese University of Hong Kong conducted comprehensive investigations using multi-cohort clinical sample analyses, Lgr5-specific transgenic mouse models, cancer cell lines, and organoid systems. The study systematically demonstrates that the m⁶A reader YTHDF1 binds to the 3′ untranslated region (3′UTR) of NOTCH1 mRNA in an m⁶A-dependent manner and promotes its translation. This regulatory axis enhances colorectal cancer stem cell stemness, accelerates tumorigenesis, and induces resistance to chemotherapy.
Importantly, the study further shows that therapeutic targeting of YTHDF1, in combination with conventional chemotherapeutic agents, markedly improves antitumor efficacy. These findings uncover a previously unrecognized molecular mechanism underlying CRC chemoresistance and propose a novel combinatorial therapeutic strategy to overcome treatment resistance in colorectal cancer.

Research Background
The clinical management of colorectal cancer (CRC) remains highly challenging. Approximately 38.5% of patients with stage II–III disease experience recurrence despite curative surgery followed by adjuvant chemotherapy, and the response rates to chemotherapy in metastatic CRC remain suboptimal. Conventional chemotherapeutic agents primarily target rapidly proliferating tumor cells; however, cancer stem cells (CSCs), endowed with self-renewal capacity, can evade chemotherapy-induced cytotoxicity and serve as the “seed cells” for tumor recurrence. N⁶-methyladenosine (m⁶A) modification regulates mRNA splicing, translation, and degradation through the coordinated actions of “writers,” “erasers,” and “readers,” and plays multifaceted roles in tumorigenesis. YTHDF1, a key m⁶A reader, has been implicated in CRC initiation and progression. Nevertheless, its CSC-specific functions, in vivo regulatory mechanisms, and contribution to chemoresistance remain poorly defined, particularly in the context of the tumor microenvironment. Therefore, elucidating the regulatory network of YTHDF1 in colorectal cancer stem cells is of critical importance for the development of novel targeted therapeutic strategies.
Research Objectives
This study aims to define the role of the m⁶A reader YTHDF1 in maintaining colorectal cancer stemness and mediating chemoresistance, to elucidate the molecular mechanisms by which YTHDF1 regulates CRC initiation and progression through downstream target genes, and to evaluate the therapeutic potential of targeting YTHDF1 in combination with chemotherapy for CRC treatment.
Research Methods
- Multi-cohort validation: Six independent clinical cohorts (N = 184, 151, 550, etc.) were analyzed. Tissue microarrays (TMAs), qPCR, Western blotting, and public database analyses were used to assess the correlations between YTHDF1 expression and CSC markers (CD133, LGR5) as well as NOTCH1, and to evaluate the association between YTHDF1 expression and chemotherapy response.
- Generation of transgenic mouse models: Lgr5-specific Ythdf1 knock-in (Ythdf1^cki^) and knock-out (Ythdf1^cko^) mouse models were established and crossed with Apc^Min/+^ mice to generate spontaneous CRC models, or subjected to AOM/DSS treatment to induce chemically driven colorectal carcinogenesis.
- Cellular models: Colorectal cancer stem cell lines (CSC28, LS174TS, POP66) and HEK293T cells were used. YTHDF1 was overexpressed or silenced, and m⁶A-binding-deficient YTHDF1 mutants (K395A/Y397A) were constructed to determine m⁶A dependency.
- Functional assays: Limiting dilution assays (LDA) were performed to evaluate stem cell self-renewal capacity. Flow cytometry was used to assess apoptosis. Western blotting was conducted to detect CSC markers, components of the NOTCH1 signaling pathway, and DNA damage markers (p-H2AX).
- Mechanistic studies: MeRIP-qPCR, RIP-qPCR, and RNC-qPCR were employed to verify the binding of YTHDF1 to m⁶A-modified NOTCH1 mRNA and its role in translational regulation. Dual-luciferase reporter constructs containing the wild-type or m⁶A-site-mutated NOTCH1 3′UTR were generated to confirm regulatory specificity.
- Pharmacological intervention: VNP-encapsulated siYTHDF1 (nanoparticle delivery system) or the YTHDF1-specific inhibitor salvianolic acid C (SAC) were applied in cell lines, organoids, and mouse xenograft models to evaluate therapeutic efficacy, either alone or in combination with oxaliplatin (OXA) and/or 5-fluorouracil (5-FU).
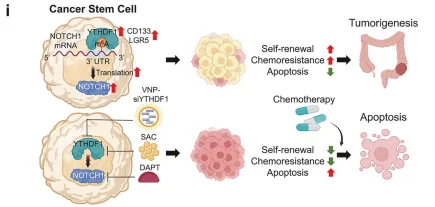
Mechanism of YTHDF1-induced CSC chemoresistance
Research Workflow
- Clinical Correlation Analysis: YTHDF1 expression was positively correlated with colorectal cancer stem cell (CSC) markers (CD133, LGR5) and was associated with chemoresistance in CRC patients. These findings were validated across multiple independent clinical cohorts, The Cancer Genome Atlas (TCGA) datasets, and single-cell RNA sequencing (scRNA-seq) analyses.
- Functional Validation: Gain- and loss-of-function studies demonstrated that YTHDF1 overexpression enhanced colorectal CSC stemness, promoted tumorigenesis, and conferred resistance to chemotherapy, whereas YTHDF1 silencing reversed these phenotypes. These functional effects were confirmed in CSC cell lines, patient-derived organoids, and Lgr5-specific transgenic mouse models.
- Mechanistic Elucidation: Mechanistically, YTHDF1 binds to the 3′ untranslated region (3′UTR) of NOTCH1 mRNA in an m⁶A-dependent manner, promoting its translation and activating the NOTCH signaling pathway. This activation suppresses apoptosis and DNA damage while reinforcing stemness properties and chemoresistance. The molecular mechanism was substantiated through integrated multi-omics analyses (MeRIP-seq, RNA-seq, Ribo-seq), protein–RNA interaction assays, and dual-luciferase reporter assays.
- Therapeutic Validation: VNP-encapsulated siYTHDF1 (nanoparticle-based delivery system) or the YTHDF1-specific inhibitor salvianolic acid C (SAC) effectively suppressed YTHDF1 activity. In combination with oxaliplatin or 5-fluorouracil, these interventions significantly inhibited tumor growth and enhanced chemotherapeutic efficacy in CRC cell models, xenograft mouse models, and patient-derived organoids.
- Clinical Translation: YTHDF1 holds promise as a prognostic biomarker for predicting chemotherapy response in colorectal cancer. Targeting YTHDF1 in combination with standard chemotherapy represents a novel therapeutic strategy to overcome chemoresistance in CRC, supported by clinical cohort correlation analyses and treatment safety evaluations.
Key Findings
-
YTHDF1 Is Positively Correlated with Colorectal Cancer Stemness
In clinical specimens, both mRNA and protein expression levels of YTHDF1 were significantly positively correlated with CSC markers CD133 and LGR5 (P < 0.001). Notably, YTHDF1 was highly enriched in the CD133⁺/LGR5⁺ CSC subpopulation. Functional assays demonstrated that YTHDF1 overexpression markedly enhanced CSC self-renewal capacity, tumorsphere formation efficiency, and in vivo tumor-initiating potential. In contrast, YTHDF1 knockdown or mutation of its m⁶A-binding domain completely abrogated these effects, confirming that YTHDF1-mediated maintenance of stemness is dependent on its m⁶A recognition function.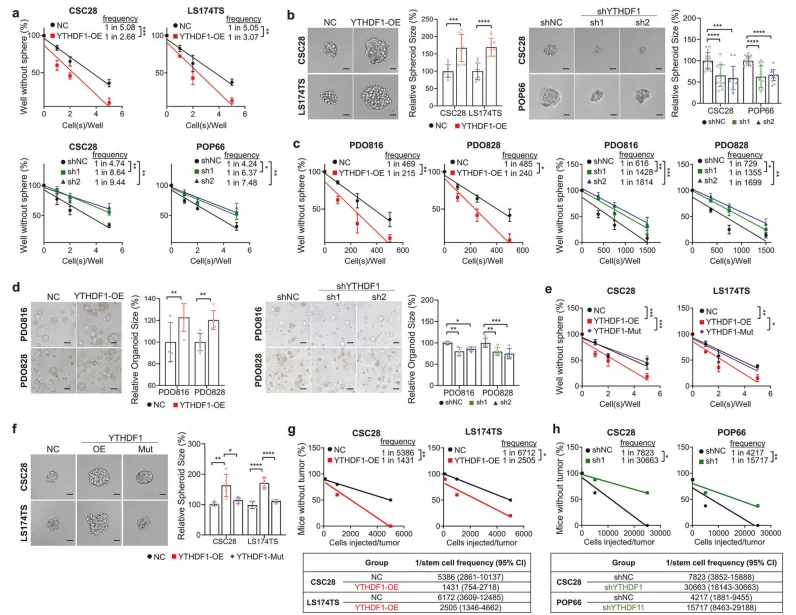
Figure 1. YTHDF1 promotes self-renewal capacity in an m⁶A-dependent manner.
-
YTHDF1 Regulates Colorectal Tumorigenesis via the m⁶A–NOTCH1 Axis
Integrated multi-omics analyses identified NOTCH1 as a critical downstream target of YTHDF1. The 3′ untranslated region (3′UTR) of NOTCH1 mRNA harbors m⁶A modification sites, to which YTHDF1 directly binds, thereby enhancing NOTCH1 translational efficiency without altering its mRNA abundance. In vivo studies further substantiated this regulatory axis. Lgr5-specific Ythdf1 knock-in (Ythdf1^cki^) mice exhibited a significant increase in tumor number and tumor burden (P < 0.05), accompanied by upregulation of canonical NOTCH downstream targets, including HES1 and HEY1. Conversely, Ythdf1 knock-out (Ythdf1^cko^) mice displayed the opposite phenotype, with reduced tumorigenesis and attenuated NOTCH pathway activation. Importantly, pharmacological inhibition of NOTCH signaling using the γ-secretase inhibitor DAPT markedly suppressed tumor development in Ythdf1^cki^ mice (P < 0.01), confirming that YTHDF1-driven tumorigenesis is mediated through NOTCH pathway activation.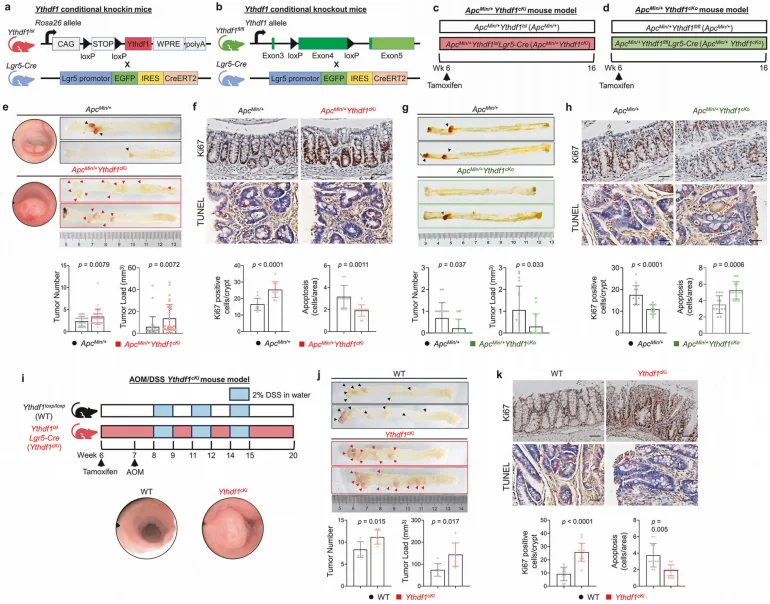
Figure 2. Lgr5-specific conditional knock-in of Ythdf1 accelerates colorectal tumorigenesis.
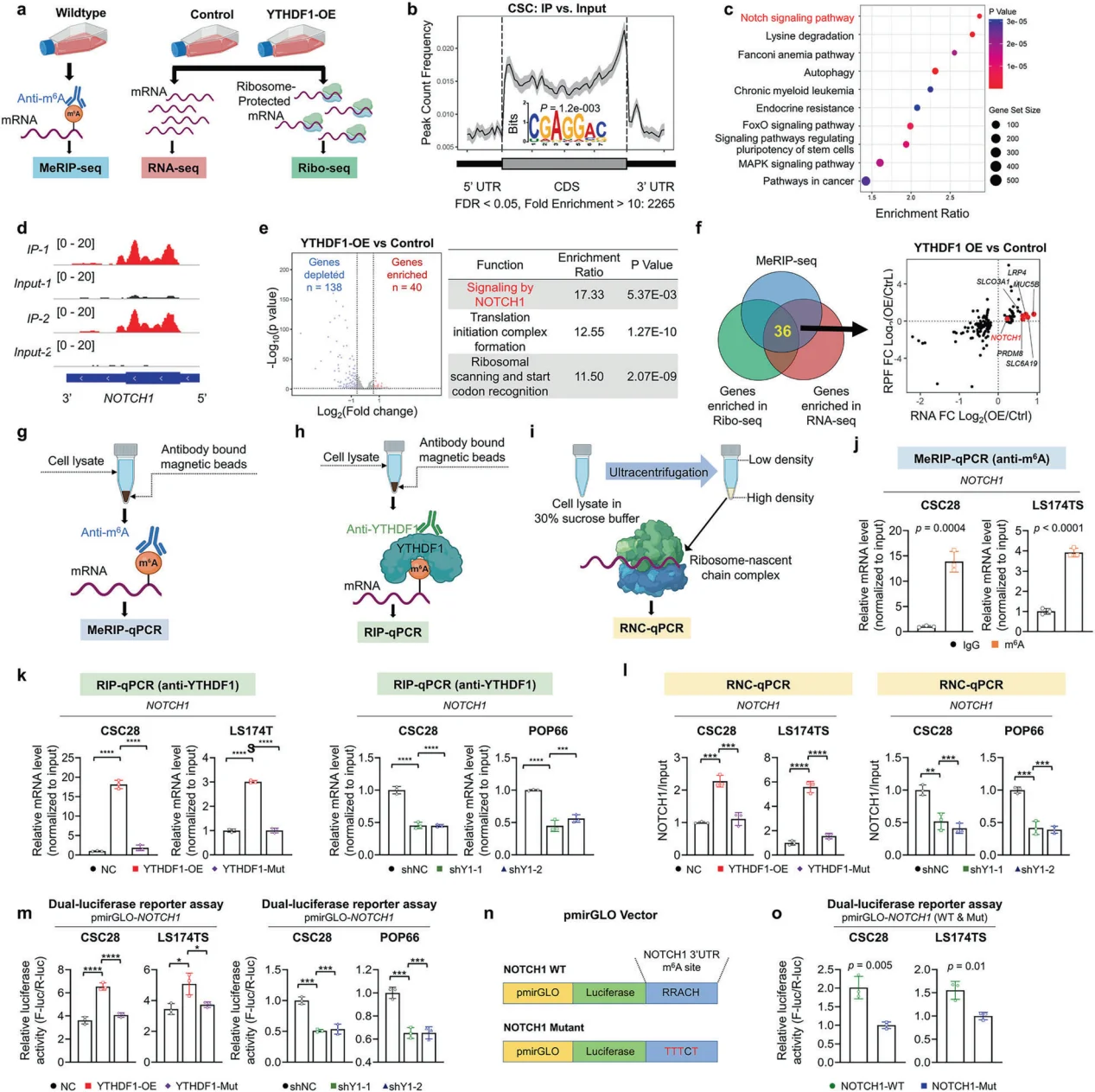
Figure 3. Integrated multi-omics analyses identify NOTCH1 as a downstream target of YTHDF1.
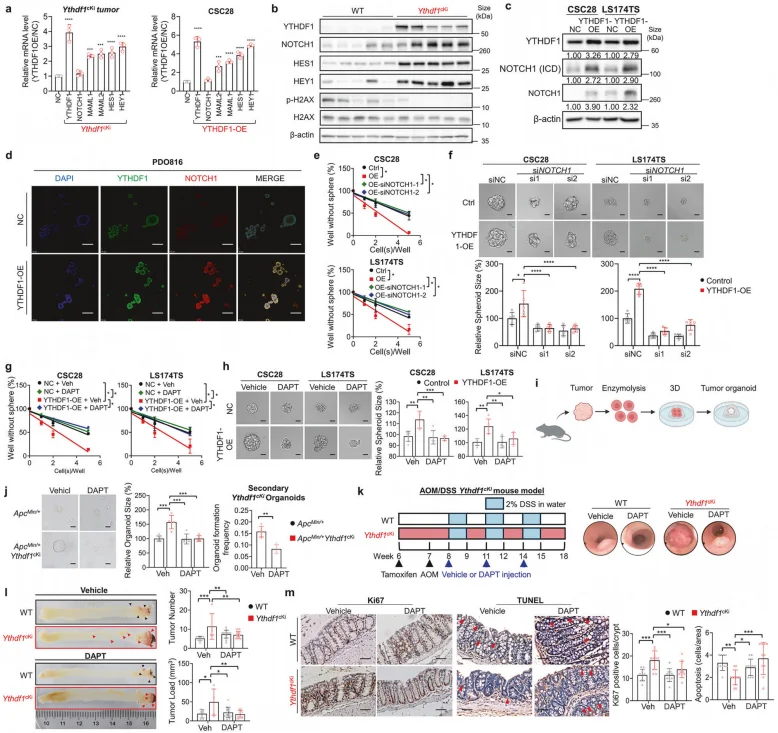
Figure 4. NOTCH1 is a functional effector mediating YTHDF1-driven stemness and tumorigenesis.
-
YTHDF1 Promotes Chemoresistance in Colorectal Cancer
In vitro, YTHDF1 overexpression significantly increased the IC₅₀ values of oxaliplatin (OXA) and 5-fluorouracil (5-FU) in colorectal CSCs and patient-derived organoids. Mechanistically, YTHDF1 attenuated chemotherapy-induced apoptosis and reduced DNA damage, as evidenced by decreased p-H2AX levels. In contrast, YTHDF1 knockdown markedly enhanced chemosensitivity and potentiated cytotoxic responses to OXA and 5-FU. In vivo, Lgr5-specific Ythdf1 knock-in (Ythdf1^cki^) mice exhibited complete resistance to OXA and 5-FU treatment, with no significant suppression of tumor growth observed. Conversely, wild-type mice displayed a significant reduction in tumor number and tumor burden following chemotherapy (P < 0.001).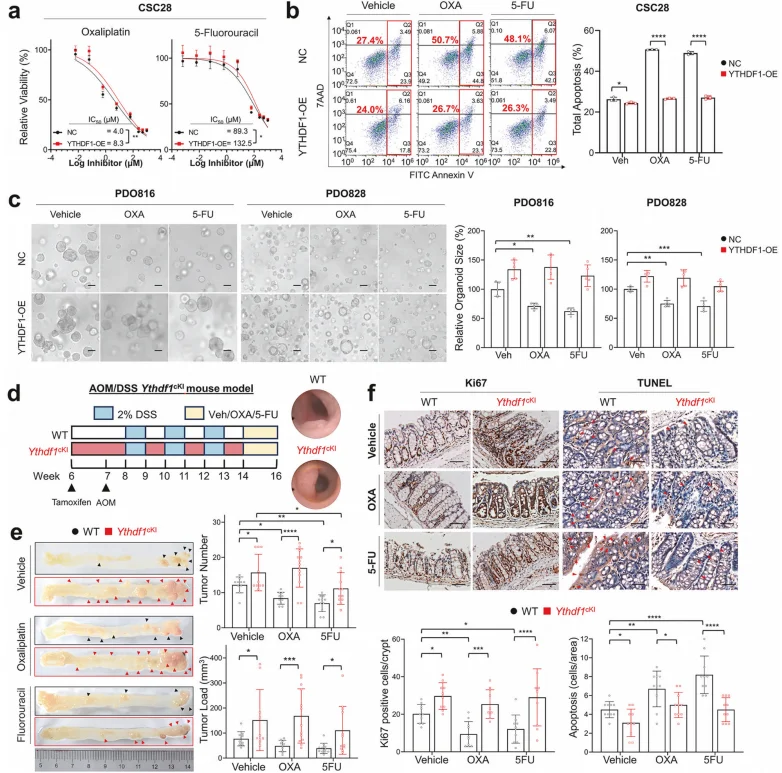
Figure 5. YTHDF1 overexpression promotes chemoresistance in colorectal cancer.
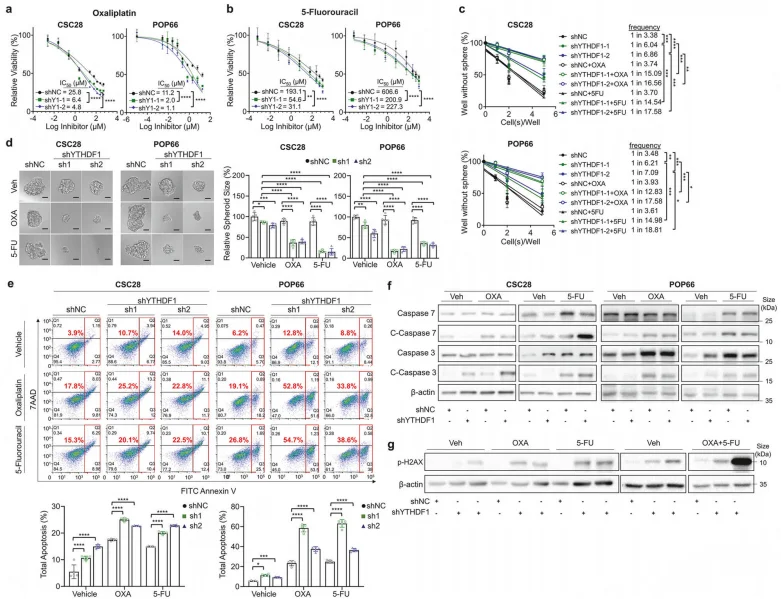
Figure 6. YTHDF1 knockdown overcomes chemoresistance in colorectal cancer.
-
Targeting YTHDF1 Enhances Chemotherapy Efficacy
In xenograft models, treatment with VNP-encapsulated siYTHDF1 alone significantly suppressed tumor growth (P < 0.05). When combined with oxaliplatin (OXA) or 5-fluorouracil (5-FU), tumor inhibition was markedly enhanced (P < 0.01), without observable hepatotoxicity or nephrotoxicity. Similarly, the YTHDF1-specific inhibitor salvianolic acid C (SAC) significantly impaired CSC stemness and organoid growth. Combination treatment with chemotherapy synergistically increased apoptosis and downregulated NOTCH1 expression, further validating the therapeutic potential of targeting YTHDF1 in CRC.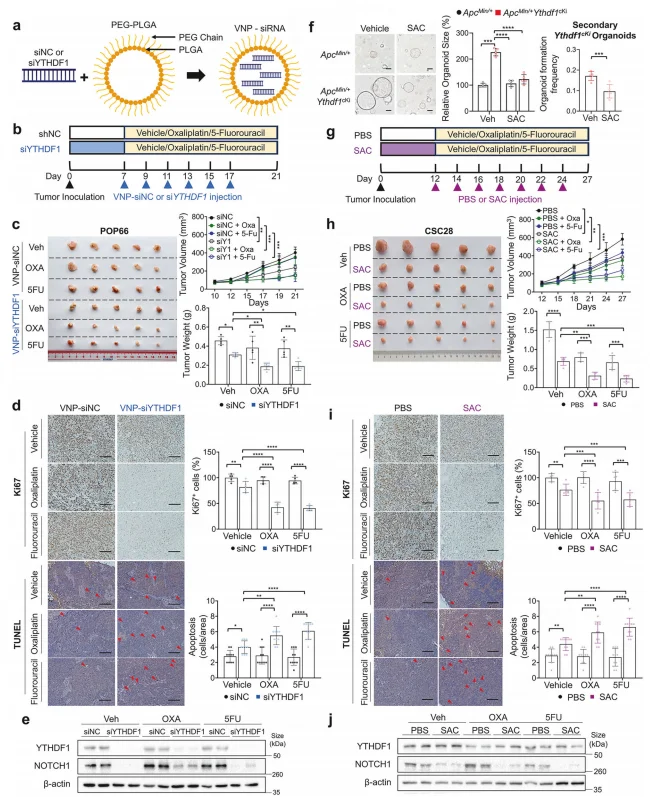
Figure 7. Chemotherapy efficacy is enhanced by targeting YTHDF1 using VNP-siYTHDF1 or salvianolic acid C (SAC).
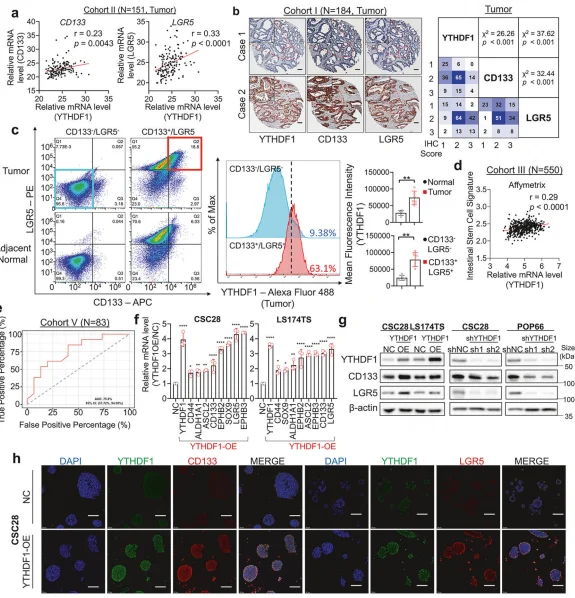
Figure 8. YTHDF1 expression positively correlates with CSC markers in colorectal cancer patient cohorts.
Significance and Innovations
-
Theoretical Significance
This study is the first to demonstrate the CSC-specific regulatory role of YTHDF1 using Lgr5-specific transgenic mouse models, revealing the core mechanism of the “m⁶A–YTHDF1–NOTCH1” axis in maintaining colorectal cancer stemness. It enriches the theoretical framework of m⁶A modifications in tumor stem cell regulation. Moreover, the discovery that YTHDF1 regulates NOTCH1 translation to modulate DNA damage repair provides a novel molecular explanation for chemoresistance in colorectal cancer. -
Clinical Significance
YTHDF1 can serve as a potential predictive biomarker for chemotherapy response in colorectal cancer, guiding clinical treatment decisions. Targeting YTHDF1 through VNP-siYTHDF1 nanoparticle delivery or the natural inhibitor salvianolic acid C (SAC) offers a safe and effective strategy to overcome chemoresistance, with SAC exhibiting strong translational potential due to its natural origin. -
Technical Innovations
This study integrates MeRIP-seq, RNA-seq, and Ribo-seq multi-omics technologies to precisely identify YTHDF1 downstream targets and regulatory patterns. The use of Lgr5-specific transgenic mouse models allows in vivo, CSC-specific modulation of YTHDF1 expression, overcoming the limitations of conventional cell line experiments that lack the native tumor microenvironment.
Summary of the Study
Through multi-cohort clinical sample analysis, Lgr5-specific transgenic mouse models, cell lines, and organoid experiments, this study systematically establishes the pivotal role of the m⁶A reader YTHDF1 in maintaining colorectal cancer stemness and mediating chemoresistance. Mechanistically, YTHDF1 binds the 3′UTR of NOTCH1 mRNA in an m⁶A-dependent manner, enhances NOTCH1 translation, activates the NOTCH signaling pathway, and thereby promotes CSC self-renewal while inhibiting chemotherapy-induced DNA damage and apoptosis. Targeting YTHDF1 effectively reverses chemoresistance, and combination therapy with oxaliplatin (OXA) or 5-fluorouracil (5-FU) significantly improves treatment efficacy. This research uncovers a novel molecular mechanism of chemoresistance in CRC and provides a theoretical basis and potential drug target for precision therapy.
Ubigene has always adhered to the core philosophy of “Make genome editing easier”, continuously iterating its products and services. To date, Ubigene has achieved over 13,000 successful gene-editing cases and offers more than 11,000 cell products (including 8,000+ knockout cell lines). With proprietary innovative technologies, Ubigene has increased gene-editing efficiency by 10–20× compared to traditional methods. Our high-quality gene-editing services and products have supported over 10,000 life science laboratories, pharmaceutical companies, and CROs worldwide.
YTHDF1 is a key m⁶A “reader” that directly regulates the translation efficiency of target mRNAs. Numerous studies have confirmed YTHDF1 as an effective therapeutic target in human cancer immunotherapy. For researchers interested in studying YTHDF1, Ubigene provides YTHDF1 knockout cell lines in popular models, including HeLa and HEK293. For customized gene-editing needs, we welcome inquiries and consultations!
Contact us for more information >>>Reference
Cheung H, Chen H, Chen D, Zhou H, Liang C, Liu W, Cheung AH, Ding Y, Yuan K, Li X, Zhang Y, Wang S, Kang W, To KF, He H, Wong CC, Yu J. YTHDF1 targets the chemotherapy response by suppressing NOTCH1-induced stemness in colorectal cancer. Signal Transduct Target Ther. 2025 Dec 22;10(1):409. doi: 10.1038/s41392-025-02507-1. PMID: 41423446; PMCID: PMC12719390.



