CRISPR Screening Breakthrough: LCN2 as a Target for Cancer Immunotherapy


Introduction
The activating transcription factor 4 (ATF4) is a central regulator of integrated stress response (ISR) activation in cancer cells. However, whether the ISR–ATF4 axis influences anti-tumour immune responses remains mostly unknown. A recent Nature study from the teams of Thales Papagiannakopoulos and Shohei Koide at New York University found that loss of ATF4 decreases tumour progression considerably in immunocompetent mice. Through CRISPR screening they identified lipocalin 2 (LCN2) as a central regulator in immunosuppression. By recruiting immunosuppressive interstitial macrophages and limiting T-cell infiltration, LCN2 is associated with an immune-excluded phenotype in patients with lung and pancreatic cancer. Anti-LCN2 antibodies can effectively stimulate anti-tumor T-cell responses, offering a new therapeutic direction for treatment-resistant solid tumors.

Background
Immune checkpoint inhibitors (ICIs) have limited efficacy in most solid tumors, and the mechanisms of tumor immune escape remain to be elucidated. Stressors in the solid-tumor microenvironment, such as hypoxia and nutrient deprivation, can activate the ISR pathway and regulate cancer-cell survival through ATF4, but the role of the ISR-ATF4 axis in shaping the tumor immune microenvironment (TME) has not yet been clarified. LCN2, a secreted glycoprotein, is known to participate in inflammatory responses, but its function in tumor immunity and its relationship with the ISR-ATF4 axis remain unknown.
Objectives
- Define the regulatory role of the ISR-ATF4 axis in anti-tumor immune responses;
- Identify the key effector molecule mediating ATF4-dependent immunosuppression;
- Elucidate the molecular mechanism by which LCN2 regulates tumor immune evasion;
- Validate the clinical prognostic value of LCN2 and the therapeutic potential of anti-LCN2 antibodies.
Methods
Cell models:
Atf4/Lcn2 knockout or overexpression cell lines (KP lung cancer, B16F10 melanoma, and KPC7 pancreatic cancer cells), as well as human LCN2-chimeric KP cells;
Animal models:
immunocompetent (C57BL/6J) and immunocompromised (NSG, nude) mice, as well as KP/KPC genetically engineered mice, used for transplantation and tumorigenesis experiments.
CRISPR screening:
construction of a 3,240-sgRNA ATF4 target-gene library, transplantation into immunocompetent or immunocompromised mice, and identification of immunity-dependent regulatory genes by NGS;
Mechanistic validation:
ChIP-qPCR was used to verify ATF4 binding to the Lcn2 promoter; LUMICKS technology was used to detect binding between LCN2 and macrophage SLC22A17; siRNA knockdown and 3D co-culture were used to validate downstream pathways;
Clinical and translational studies:
analysis of tissue microarrays from 105 lung cancer and 33 pancreatic cancer cases, together with TCGA data to correlate LCN2 expression with prognosis; evaluation of the in vivo efficacy of anti-mLCN2 / anti-hLCN2 antibodies.
Key Findings
1. ATF4 regulates immunity-dependent tumor growth
Atf4KO suppressed tumor progression only in immunocompetent mice, and the ISR inhibitor ISRIB prolonged survival, confirming that ATF4 promotes tumor growth by suppressing anti-tumor immunity.
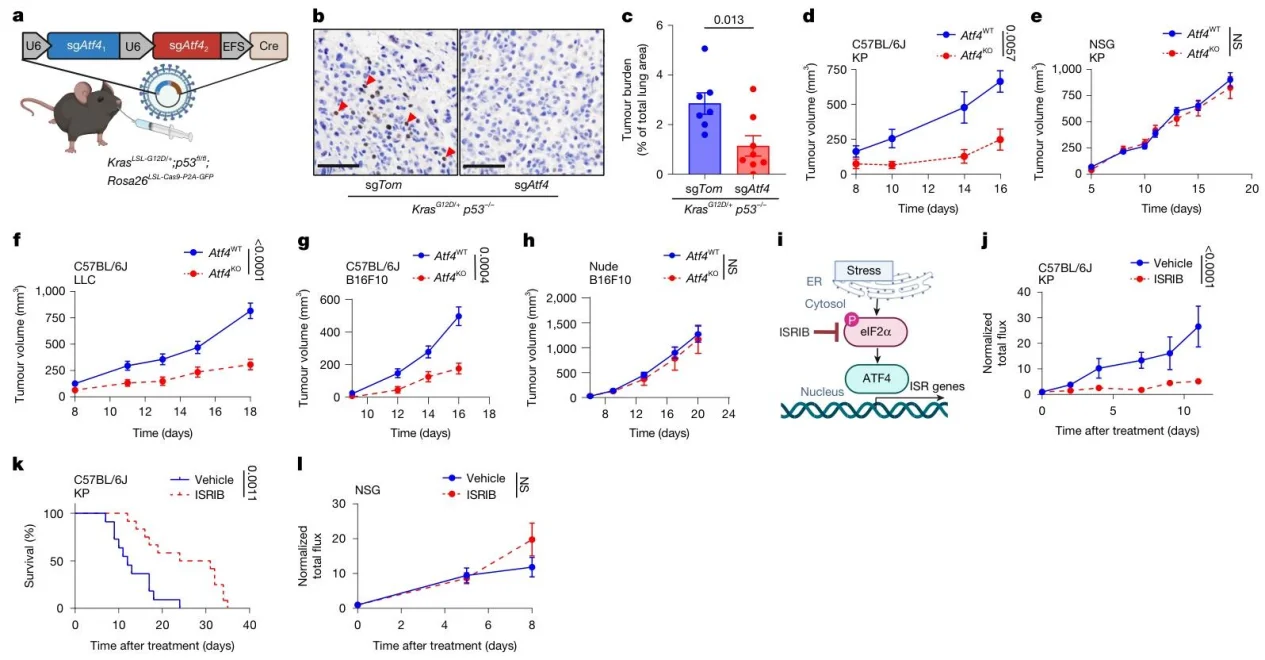
Fig.1. ATF4 is essential for tumor growth in immunocompetent mice
2. CRISPR screening identifies LCN2 as a central regulator
Lcn2-targeting sgRNAs were significantly depleted in immunocompetent mice, the Lcn2KO phenotype mirrored that of Atf4KO, and LCN2 overexpression rescued the tumor-growth defect; its function depended on the secretion signal.
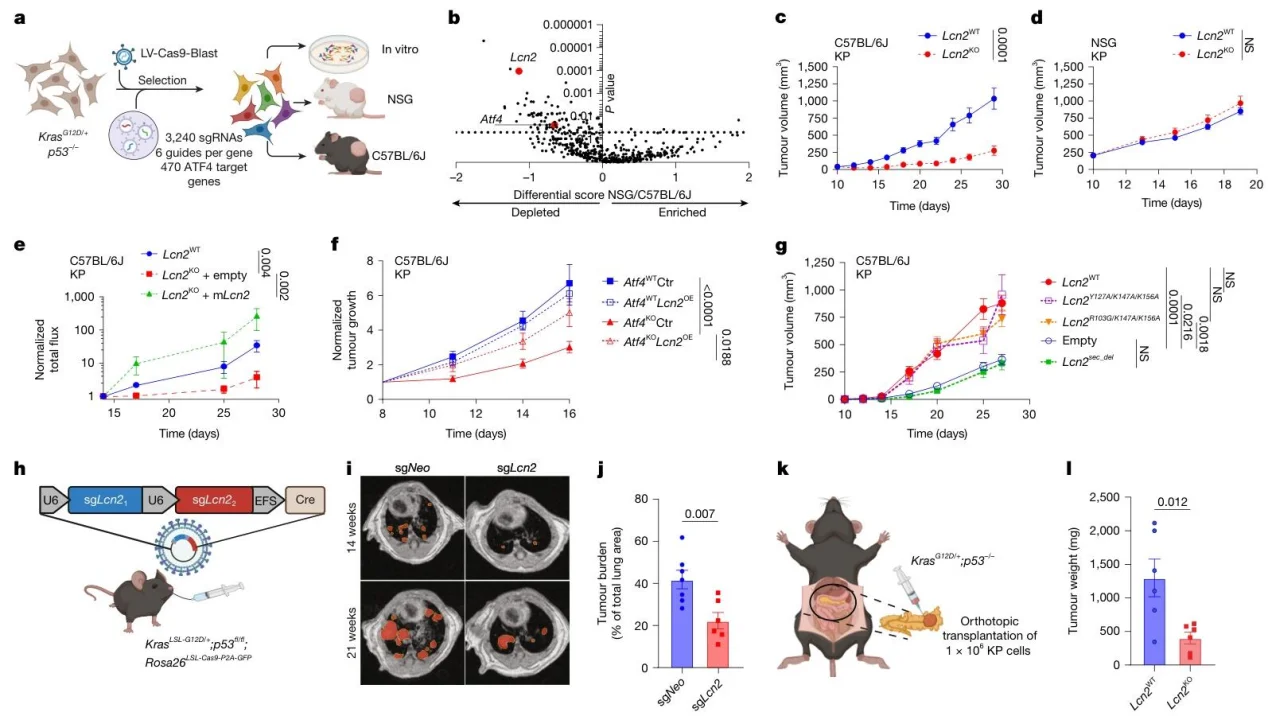
Fig. 2: LCN2 loss slows tumour progression in immunocompetent mice.
3. LCN2 suppresses T-cell responses by polarizing macrophages
Lcn2KO tumors showed increased CD4+/CD8+ T-cell infiltration and reduced Treg cells; LCN2 binds macrophage SLC22A17, suppresses Cxcl9 secretion, promotes Il6 expression, and blocks T-cell chemotaxis.
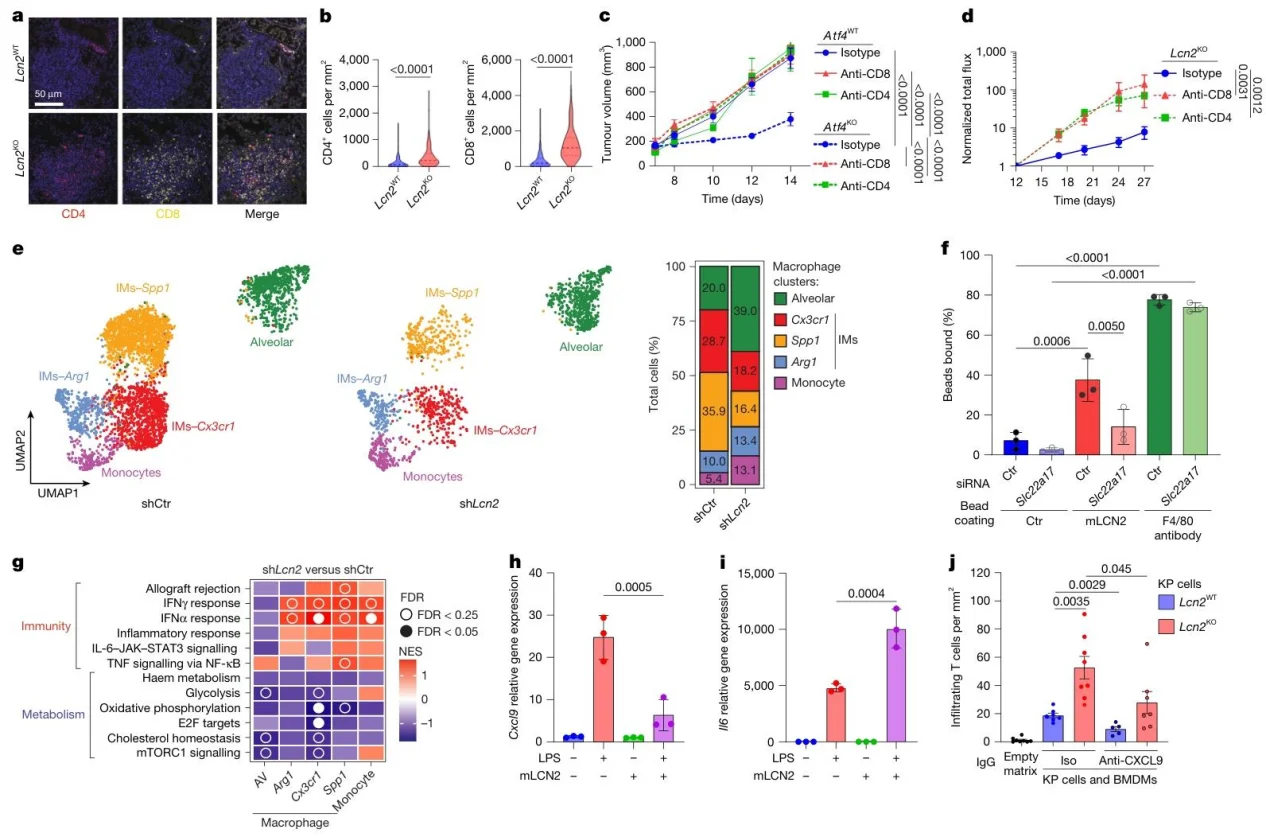
Fig.3. LCN2 promotes an immunoregulatory macrophage state that weakens impairs immune responses to cancer
4. LCN2 is associated with clinical immune exclusion and prognosis
In patients with lung and pancreatic cancer, high LCN2 expression was associated with reduced CD3+ T-cell infiltration and higher tumor grade; TCGA data showed a strong association with the immune-excluded phenotype and poor prognosis.
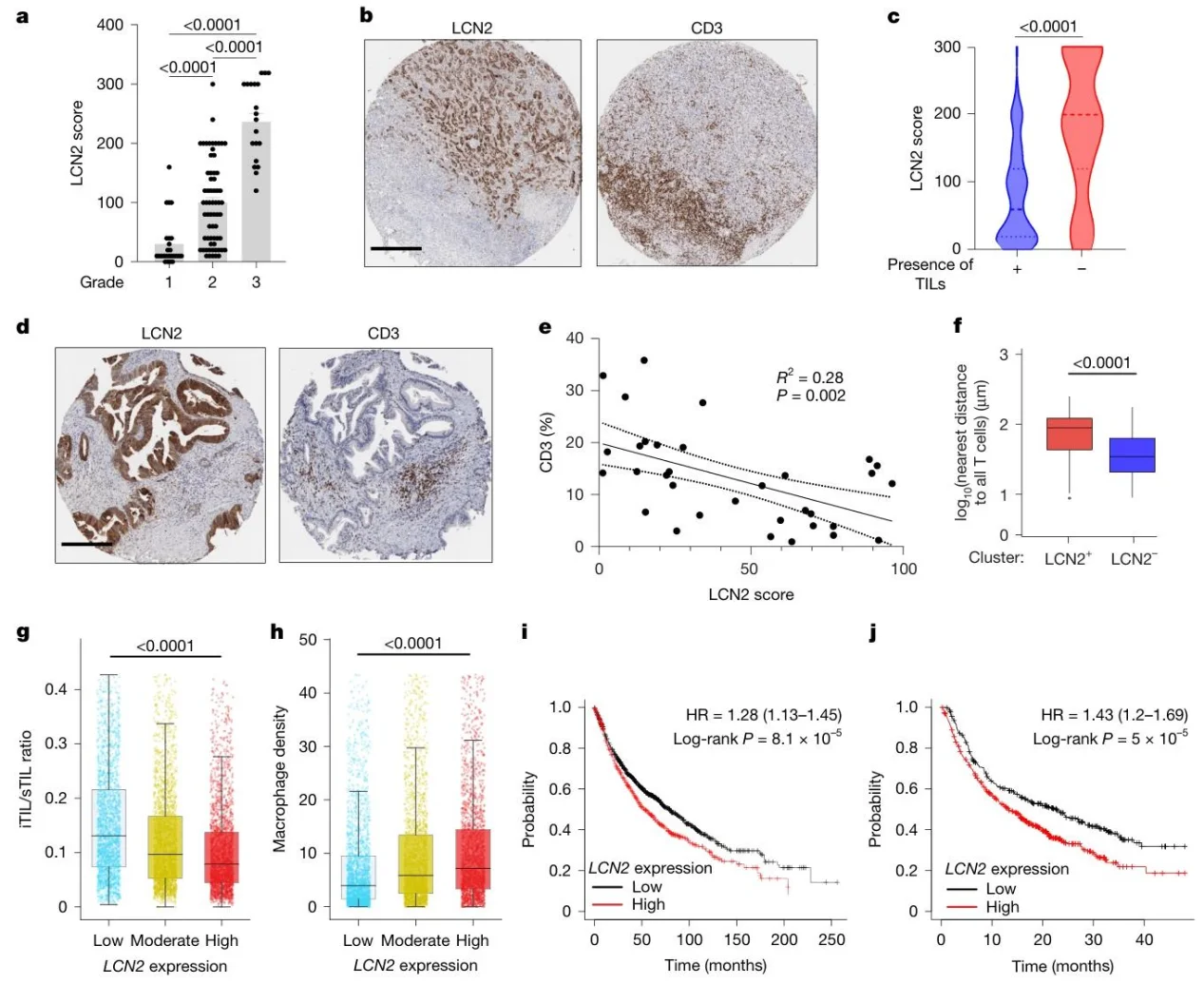
Fig. 4: LCN2 levels in patients correlate with higher tumour grade and reduced TILs.
5. Anti-LCN2 antibodies have robust efficacy in aggressive tumour models
anti-mLCN2 / anti-hLCN2 inhibited the growth of orthotopic lung and pancreatic tumors, increased T-cell infiltration, and further prolonged survival when combined with anti-PD-1 treatment, without obvious toxicity.
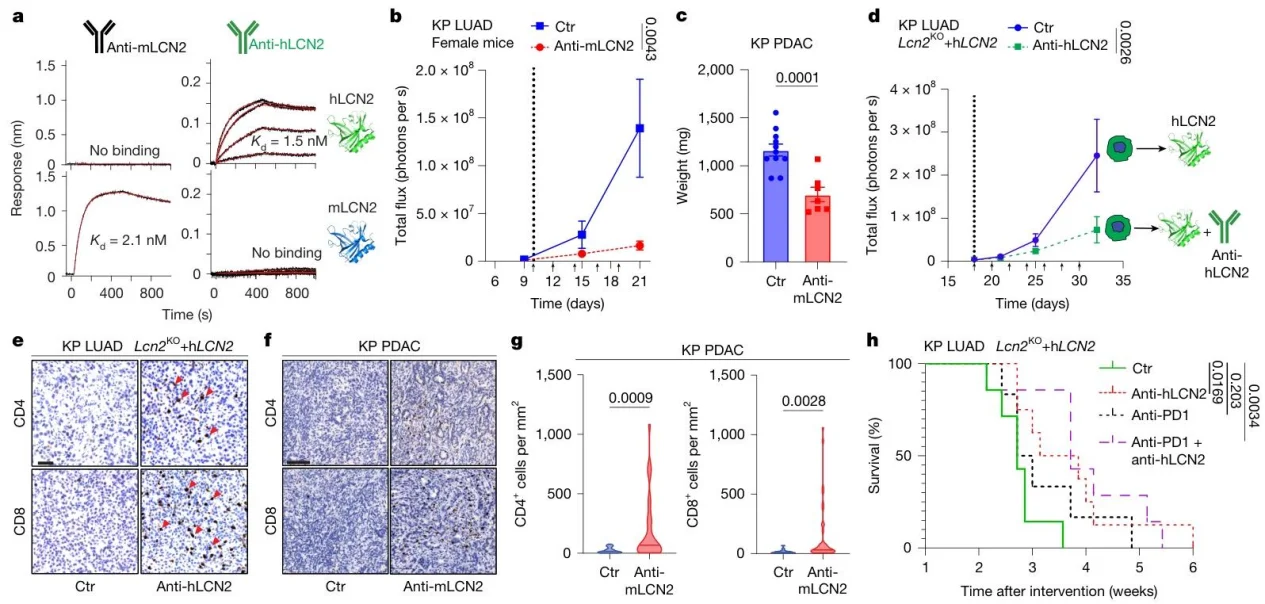
Fig. 5. Synthetic antibodies developed against LCN2 show therapeutic benefit and anti-cancer activity
Summary
This study found that ISR-ATF4 axis promotes formation of an immunosuppressive TME, suppress T-cell infiltration, and mediate immune evasion by inducing LCN2 secretion and engages the macrophage receptor SLC22A17. Clinical data showed that high LCN2 expression is associated with an immune-excluded phenotype and poor prognosis in patients with solid tumors. Anti-LCN2 antibodies can effectively block this pathway and stimulate anti-tumor immune responses, with even better results when combined with PD-1 inhibitors. The findings not only deepens our understanding of the link between stress responses and tumor immunity, but also provides a new therapeutic strategy for immune-excluded solid tumors.
CRISPR-iScreen™ Libraries from Ubigene
CRISPR-iScreen™ is an innovative technology independently developed by Ubigene, designed to achieve efficient CRISPR screening. Currently, Ubigene offers over 40 CRISPR library products in stock, covering genome-wide knockout/Interference/activation libraries for humans, mice, green monkeys, pigs, and other species. Additionally, there are CRISPR knockout/Interference/activation sub-libraries targeting genes related to kinases, the cell cycle, membrane proteins, and metabolism, helping you easily identify targets of interest! Ubigene currently has over 150 CRISPR library virus and over 600 CRISPR library Cell Pools ready to use, and also provides one-stop CRISPR (in vitro/in vivo) functional screening services to readily support your research needs.
Ubigene also provides personalized CRISPR library construction services, tailored to your screening goals and experimental objectives. We offer one-on-one consultation to help optimize screening strategies and library selection.
Contact us for more details >>>


