Nature Outlook 2026: Gene Editing Poised to Drive the Next Wave of Precision Medicine


Gene Editing Trends in 2026
Recently, Nature published a feature reviewing major scientific trends expected to shape the global research landscape in 2026. Among fields such as artificial intelligence and biomedicine, gene editing —particularly CRISPR-based gene editing —has emerged as a central driver of precision medicine . With personalized gene editing therapies for rare diseases entering clinical trials, the field is rapidly moving toward real-world clinical application.

Personalized CRISPR Therapies Enter Clinical Trials
The year 2026 could mark an important milestone for gene editing in clinical medicine. Much of the current momentum stems from a pioneering case involving a baby with a rare metabolic disorder, KJ Muldoon, whose condition was successfully stabilized following treatment with a customized CRISPR-based therapy. This landmark case has laid a solid foundation for subsequent large-scale clinical trials and demonstrated the feasibility of translating personalized gene therapy from laboratory research into patient care.
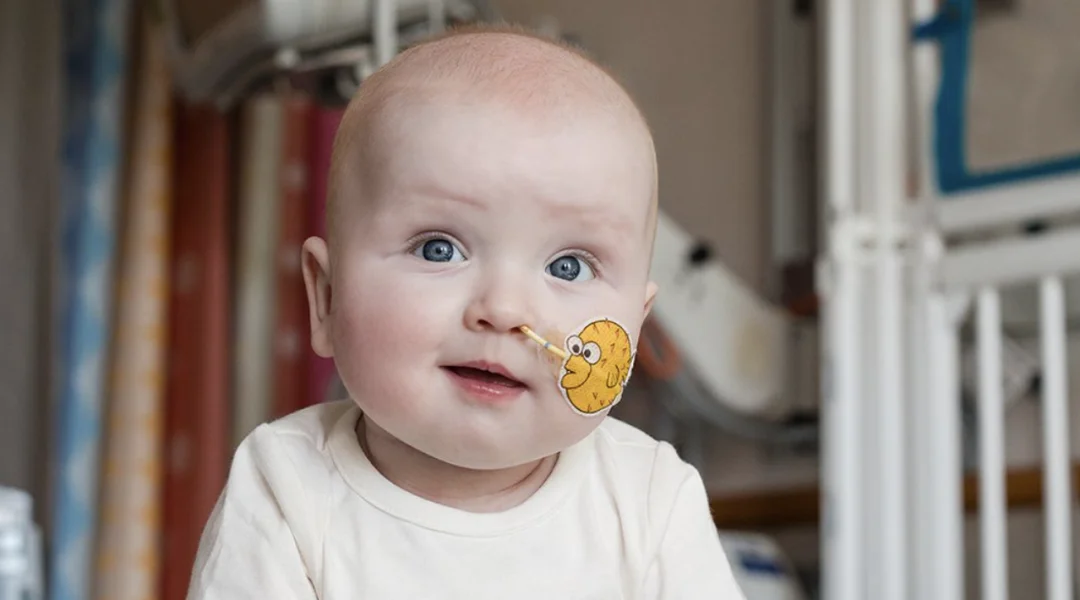
The image is sourced from Nature
According to Nature, at least two key clinical trials may begin in 2026. The research team that treated Muldoon plans to seek approval from the U.S. Food and Drug Administration (FDA) to launch a clinical trial in Philadelphia targeting children with a wider range of rare metabolic diseases. The proposed trial would include conditions caused by mutations in 7 different genes, all of which could potentially be addressed using a similar gene-editing strategy. If successful, the approach could demonstrate how a single technological framework might be adapted to treat multiple rare disorders affecting small patient populations.
Another group is preparing a personalized gene-editing trial for inherited immune-system disorders. Unlike conventional therapies that primarily manage symptoms, these approaches aim to directly correct the underlying genetic mutation, halting disease progression at its source and offering new hope for families long burdened by rare genetic diseases.
Gene editing is also advancing in cancer research. A Chinese biotechnology company developing an in vivo CAR-T therapy is expected to release its first clinical data in the second or third quarter of 2026. This approach is designed to modify T cells directly within the patient's body. directly within the patient's body. This technology enables gene modification and activation of T cells directly within the patient's body via a single intravenous injection of viral vectors, eliminating the need for ex vivo cell manufacturing. Such an approach could significantly shorten treatment timelines and reduce the high cost associated with conventional CAR-T therapies, thereby facilitating broader access to advanced cancer treatments.
Regulatory Reforms as Catalysts
Accelerating the Translation of Gene Therapies from Bench to Bedside
Progress in gene-editing therapies is occurring alongside regulatory reforms aimed at streamlining clinical research. Both the United Kingdom and the United States are introducing policy changes that could facilitate the development and approval of innovative medical technologies.

The image is sourced from internet
In the United Kingdom, the largest reformation of clinical-trial regulations in two decades will take effect in April 2026. Under the new framework, researchers will be able to obtain both ethical approval and regulatory authorization through a single application process. The reforms also require all drug-related clinical trials to be registered publicly before the first participant is enrolled and to publish summary results within 12 months of completion. The changes are expected to significantly shorten approval timelines while enhancing transparency and participant diversity, enabling promising gene-editing therapies to reach patients more rapidly.
In the United States, the FDA proposed new regulatory measures in December 2025 aimed at reducing the number of clinical trials required for new drug approval from two to one. The policy is expected to be gradually implemented in 2026 and is intended to accelerate research progress, increase participant diversity, and shorten the time required for new therapies to reach patients.
Parallel Progress Across Scientific Fields
Highlights in Artificial Intelligence and Biomedicine
Beyond gene editing, several other scientific fields are expected to advance rapidly in 2026. In artificial intelligence, AI agents that integrate multiple large language models are beginning to automate complex research workflows and could soon contribute to AI-driven scientific discoveries. Smaller AI models, with lower training costs and strong task-specific reasoning capabilities, may complement larger systems, although their limitations could become more apparent as adoption grows. In biomedicine, results are expected from a large UK clinical trial involving more than 140,000 participants evaluating a blood test designed to detect signals from up to 50 cancers from a single sample—an approach that could reshape early cancer screening.

The image is sourced from internet
Outlook
Taken together by Nature, these developments suggest that 2026 could be a pivotal year for both scientific discovery and the evolution of the research ecosystem. Gene-editing technologies are moving steadily from experimental research toward clinical application, potentially redefining treatment options for rare genetic diseases. At the same time, advances in artificial intelligence, biomedical technologies, and regulatory policy may accelerate the pace at which new scientific insights translate into real-world impact.
These advances are closely linked to the increasing demand for reliable gene editing platforms, including CRISPR knockout cell lines and high-throughput screening services.
About Ubigene
Ubigene Biotechnology Co., Ltd.(Ubigene) is an international high-tech enterprise serving the global life sciences and biopharmaceutical industries. The company was co-founded by a group of experts with extensive academic and industrial experience in gene editing and cell biology. Centered on gene-editing technologies as its core driving force, Ubigene Biotech has established an integrated technological ecosystem encompassing cutting-edge research and development, large-scale application, and industrial translation, with the goal of delivering scalable, reproducible, and industrializable solutions for life science research and biomanufacturing.
Built around CRISPR gene-editing technology as a foundational platform, Ubigene has developed internationally advanced gene-editing technology platforms, innovative cell biology platforms, and high-throughput screening systems. These capabilities form a comprehensive workflow covering design, construction, screening, analysis, and validation. Since establishment, we have provided highly consistent, stable, and scalable gene-editing products and technical services to more than ten thousand research institutions, biopharmaceutical companies, and CRO organizations worldwide. These solutions are widely applied in target discovery, functional genomics screening, drug development, and the construction of cellular models.
Leveraging our independent innovation capabilities and AI-driven research tools that integrate multiple large language models, Ubigene has developed two powerful tools: the Red Cotton·CRISPR Gene Editing System and the iScreenAnlys™ Library Analysis Platform. These tools significantly enhance the efficiency and reliability of large-scale, high-throughput, industrial-level gene-editing screening, providing robust technical support for precise regulation and data analysis in complex biological systems. This integrated technological framework substantially shortens the translation timeline from fundamental research to industrial applications, establishing a standardized foundation for large-scale screening and pipeline development.
In terms of product and service development, Ubigene has established four core product lines that systematically support diverse applications of gene-edited cells, including drug screening, functional studies, disease modeling, and biomanufacturing. We also offer comprehensive integrated services—including CRISPR gene editing, CRISPR library construction and screening, iPSC gene editing, and directed differentiation—supporting continuous needs from early-stage research exploration to industrial-scale development.
Looking ahead to 2026, Ubigene will continue to advance its integrated development strategy centered on intelligent technology platforms, core product portfolios, and customized solutions. The company aims to promote the standardization, scalability, and industrialization of gene-editing technologies, empowering the upgrading of biomanufacturing and the biopharmaceutical industry through engineering and data-driven capabilities, and accelerating the industrial implementation of key biotechnologies to contribute to a more efficient and sustainable global biopharmaceutical ecosystem.
Reference
https://www.nature.com/articles/d41586-025-03673-6



