CHO ko cell line
Introduction:
CHO cells are also called Chinese hamster ovary cells, which are an epithelial cell line that originated from the ovary of the Chinese hamster. Chinese hamster ovary (CHO) cells, with their unique characteristics, have become a major workhorse for the manufacture of therapeutic recombinant proteins. This cell line is usually used in biological and medical research. Also, it is the most widely used mammalian host in terms of the industrial production of therapeutic proteins.
Application:
Construction of a gene knockout CHO cell line using gene targeting method
Since CHO cell line is a major host for therapeutic antibody production, constructing productive CHO cell lines is important. There are two major transfection methods that are commonly used in building characterized CHO cell lines, one is random integration and the other one is gene targeting. The former one, which causes variation in antibody productivity, would usually affect transgene expression levels.
However, in gene-targeting methods, exogenous genes are inserted into a specific chromosomal region. This method is based on homologous recombination using sequences targeting a specific genomic region of the host cell. Researchers used CRISPR/Cas9 system as a gene-targeting method, which induces double-strand breaks(DSBs) via guide RNA and Cas9, which increases the efficiency of homologous recombination. Therefore, CRISPR/Cas9 vector system can efficiently insert exogenous genes into CHO cell lines.
Improving the efficiency of CHO cell line generation using glutamine synthetase gene knockout cells
Although Chinese hamster ovary (CHO) cells, with their unique characteristics, have become a major workhorse for the manufacture of therapeutic recombinant proteins, one of the major challenges in CHO cell line generation (CLG) is how to efficiently identify those rare, high-producing clones among a large population of low- and non-productive clones. Currently, there are two main CHO expression systems that have been widely used, dihydrofolate reductase (DHFR)-based methotrexate (MTX) selection and glutamine synthetase (GS)-based methionine sulfoximine (MSX) selection.
To study endogenous GS expression's potential impact on selection efficiency, GS-knockout CHOK1SV cell lines were generated using the zinc finger nuclease (ZFN) technology designed to specifically target the endogenous CHO GS gene. The high efficiency (∼2%) of bi-allelic modification on the CHO GS gene supports the unique advantages of the ZFN technology, especially in CHO cells. GS enzyme function disruption was confirmed by the observation of the glutamine-dependent growth of all GS-knockout cell lines.
Characterization of glutamine synthetase-mediated selection for the establishment of recombinant CHO cells producing monoclonal antibodies
With a GS-knockout CHO cell line12 and promoter engineering, the GS-based system can be effective in cell line generation even in the absence of MSX1. Using two different host cell lines (CHO-K1 and GS-knockout CHO (GS KO), mAb producing rCHO cell clones were generated by a single round of selection at various MSX concentrations.
GS-knockout CHO cell lines with an improved selection stringency. The use of the GS-knockout CHO host cell line facilitates the rapid generation of high producing clones with reduced production of lactate and ammonia in the absence of MSX.
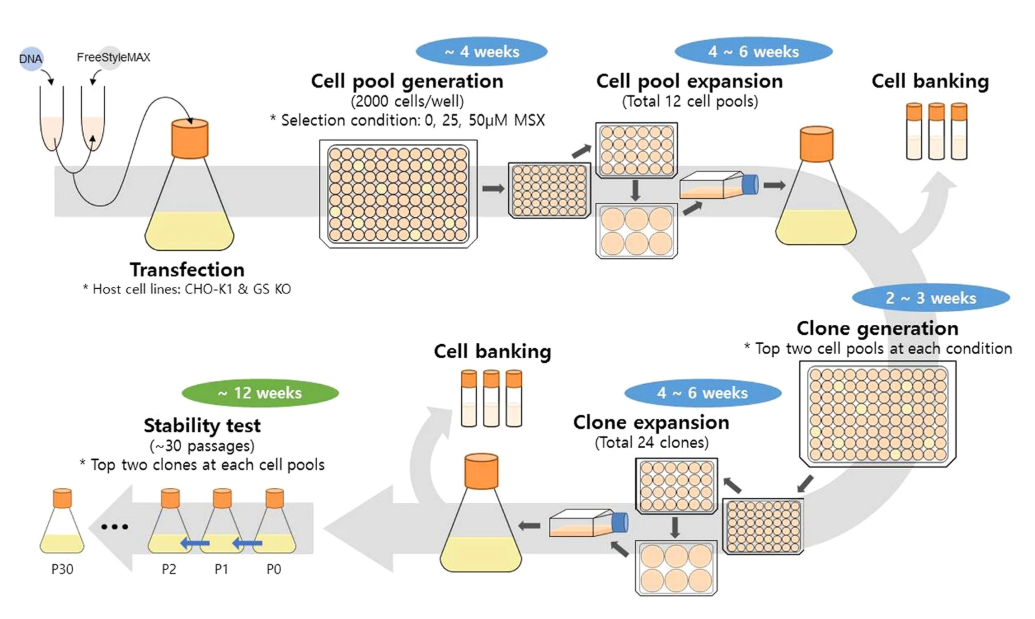
A schematic diagram of the process for mAb producing clone generation and long-term culture for testing the production stability.
CRISPR/Cas9 knockout HEK293 cell line strategies

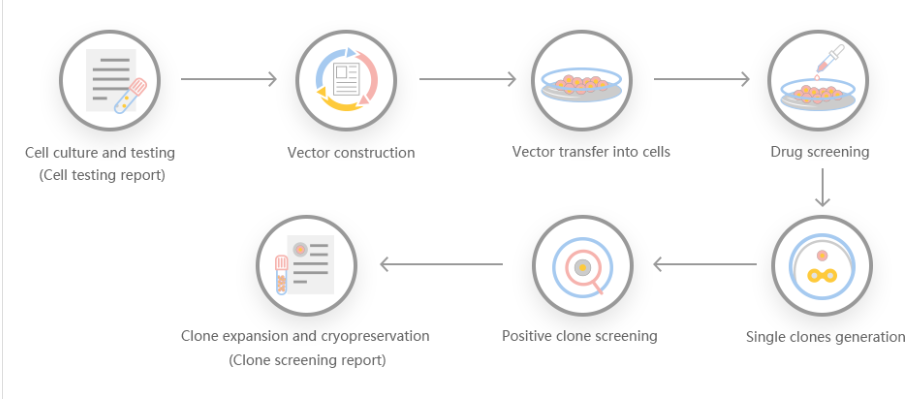
Work flow of knockout HEK293 cell line
Reference:
Fan L, Kadura I, Krebs LE, Hatfield CC, Shaw MM, Frye CC, Improving the efficiency of CHO cell line generation using glutamine synthetase gene knockout cells.
Aga, M., Yamano, N., Kumamoto, T. et al. Construction of a gene knockout CHO cell line using a simple gene targeting method. BMC Proc 9, P2 (2015).
Noh, S.M., Shin, S. & Lee, G.M. Comprehensive characterization of glutamine synthetase-mediated selection for the establishment of recombinant CHO cells producing monoclonal antibodies. Sci Rep 8, 5361 (2018).
 Subscribe Us
Subscribe Us Gene Editing Services
Gene Editing Services
 EZ-editor™
EZ-editor™ Red Cotton Gene knockout Project
Red Cotton Gene knockout Project










