Gene Knockin Cell Line
Cells are co-transfected via nucleofection with a gRNA expression vector (carry Cas9) and a donor template carrying the insert of interest. A site-specific DNA double-strand break (DSB) is introduced at the target locus, and precise knock-in is achieved through homology-directed repair (HDR) using the donor as a template. Following antibiotic selection, single-cell clones are isolated (fluorescence can be used for preliminary screening if applicable). Targeted PCR and Sanger sequencing are performed to confirm correct integration, and validated positive clones are delivered with a complete data report.
Gene Knockin Cell Line Service Detail
Service Type
Gene Knockin
Cell Line Type
Tumor Cells, Immortalized Cell Lines. Various cell types, including iPS cells
Project Price (USD)
From $5980
Turnaround
As fast as 10 weeks
Deliverables
1. ≥1 homozygous Single Clone(>106 cells/vial, 2 vials per clone) 2. Wild-Type Cells(>106 cells/vial, 2 vials) 3. Experimental Reports
Knockin Validation
PCR(Standard), Sanger sequencing(Standard)
Digestive System
Endocrine System
Respiratory System
Reproductive System
Circulatory System
Blood and lymphatic System
Brain and Nervous System
Urinary System
Skeleton, Articulus, Soft Tissue, Derma System
Stem Cell Lines
Ocular, Otolaryngologic and Oral System
Gene Knockin (KI) Cells Construction Methods
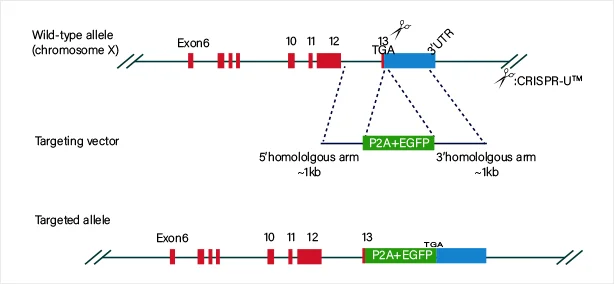
Protein fusion
View Picture
The guide RNA and Cas9 complex induce a double-strand break (DSB) at the target site of the DNA. The donor vector carrying the knock-in sequence serves as the template for homologous recombination repair (HDR), facilitating the recombination of the knock-in sequence into the target site.
Replacement of specific locus
View Picture
Workflow and Validation
Strategy Design by Red Cotton System

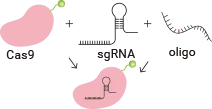
RNP Complex
Cell Transfection

Pool Efficiency Validation
Single-cell Cloning
PCR Amplification
Sanger Sequencing Validation
QC & Cell Cryopreservaion

Case Study
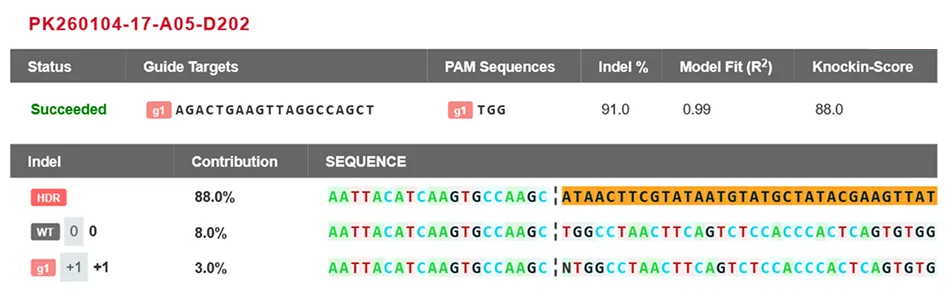
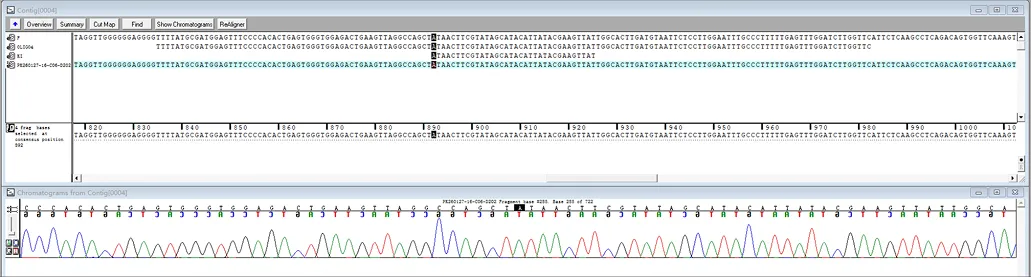
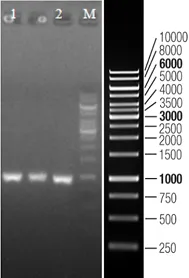
Research Review
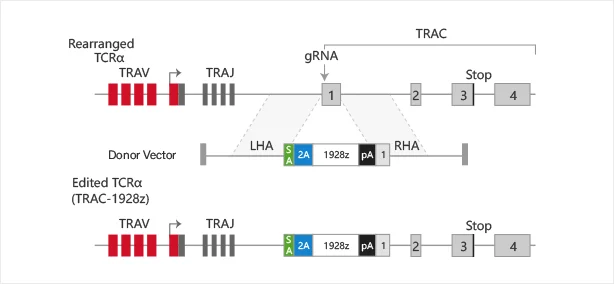
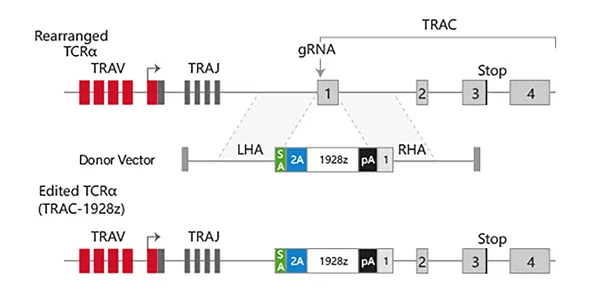
Targeting a CAR to the TRAC locus with CRISPR/Cas9 enhances tumour rejection
T cells expressing chimeric antigen receptors (CARs) can mediate tumor rejection and are an effective therapy for B-cell malignancies. By applying site-specific genome editing, a CD19-specific CAR cassette can be precisely integrated into the TRAC locus in T cells via donor template–mediated homologous recombination. This strategy enables uniform CAR expression in primary human peripheral blood T cells and enhances their immune response capacity.
View Picture
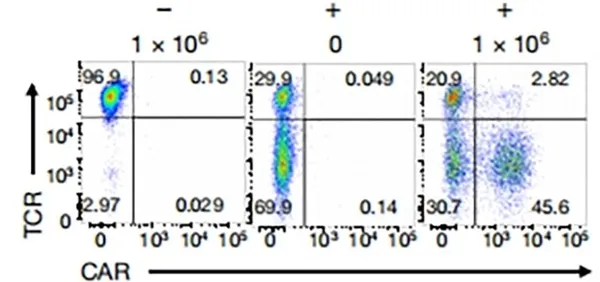
View Picture
FAQs
1. What is CRISPR knock-in?
CRISPR knock-in refers to the process of using the CRISPR/Cas9 gene-editing technology to insert a specific genetic sequence (such as a gene, regulatory element, or mutation) into a precise location in the genome. This technique is used to add functional genes or introduce specific genetic modifications in a targeted manner, as opposed to CRISPR knock-out, where genes are disrupted or deleted.
2. How does CRISPR Knock-in Work?
- Designing the Guide RNA (gRNA): A guide RNA is designed to specifically target a location in the genome where the new DNA sequence will be inserted. This RNA sequence is complementary to the target DNA sequence, ensuring that Cas9 cuts the correct location in the genome.
- Cas9 Protein: The Cas9 protein, which is part of the CRISPR system, is responsible for making a double-strand break at the target location in the DNA.
- Donor DNA Template: To achieve the knock-in, a donor DNA template is introduced into the cell. This template contains the genetic sequence that will be inserted into the genome. The donor template typically has homology arms (sequences that match the regions adjacent to the target break) to guide the cell's repair machinery.
- Homology-Directed Repair (HDR): After Cas9 cuts the DNA, the cell uses its HDR mechanism to repair the break. If a donor template is provided, the cell incorporates the new genetic material from the donor DNA into the genome at the break site, which results in the insertion of the desired sequence.
3. What are the differences between CRISPR gene knock-in and knock-out ?
| key Differences | ||
|---|---|---|
| Feature | CRISPR Knock-in | CRISPR Knock-out |
| Purpose | To insert a specific gene or sequence into the genome. | To disrupt or inactivate a gene in the genome. |
| Outcome | Addition of a gene or sequence at a specific site. | Loss-of-function mutation due to gene disruption. |
| Repair Mechanism | Homology-directed repair (HDR) using a donor template. | Non-homologous end joining (NHEJ) that introduces errors at the break site. |
| Resulting Change | Insertion of new genetic material (e.g., a gene, mutation, or reporter sequence). | Gene is disrupted or inactivated, often with indels. |
| Use Cases | Gene therapy, disease modeling, synthetic biology, protein production. | Gene function studies, disease modeling, cancer research, drug discovery. |
4. What are the applications of CRISPR Knock-in?
- Gene Therapy: Knock-in CRISPR can be used to insert healthy copies of defective genes in patients with genetic disorders. For example, correcting mutations in the CFTR gene (responsible for cystic fibrosis) by inserting a functional copy into a patient's cells.
- Disease Modeling: Researchers use knock-in to introduce specific mutations into the genome of animal models (like mice or rats) to study diseases or test therapies. This helps in creating models of genetic diseases such as cancer or neurodegenerative conditions.
- Synthetic Biology: Scientists can insert new genes into organisms (bacteria, yeast, or plants) to create new functions, such as engineered microbes that produce biofuels or therapeutic proteins.
- Drug Development: In drug discovery, knock-in CRISPR is used to create cell lines or animals that express specific mutations or diseases, allowing for more accurate testing of drugs or potential treatments.
- Cell Line Engineering: For industrial or therapeutic use, CRISPR knock-in can generate cell lines that express desired proteins, enzymes, or other bioactive molecules for research or commercial purposes.
5. How to perform a CRISPR Knockin Experiment?
- Cell Type: Choose an appropriate cell line or primary cell type for your experiment.
- Efficiency: CRISPR knock-ins generally have lower efficiency than knock-outs, and larger inserts can reduce efficiency further.
- Delivery Method: Choose a delivery method (electroporation, lipid-based, viral) based on your cell type's transfection efficiency.
- Homology-directed Repair (HDR): The success of knock-ins heavily relies on HDR, which is not as efficient as non-homologous end joining (NHEJ). You can enhance HDR by using HDR enhancers or small molecules (like SCR7).
- Verification: Always validate your knock-in using multiple methods (PCR, sequencing, protein expression).
Ubigene could provide 4 solutions to meet different mutation needs! Get in touch with our experts now
6. How to validate CRISPR knockin cell?
Validating CRISPR knock-in cells involves confirming:
- Correct insertion of the desired gene at the target location (via PCR, sequencing, Southern blot).
- Expression of the inserted gene at the RNA and protein levels (via RT-PCR, Western blot, fluorescence assays).
- Functionality of the inserted gene, especially for functional knock-ins (via fluorescence or activity assays).
- Absence of off-target effects, to ensure the edit is precise (via sequencing or specific assays).
7. What are the challenges and limitations of CRISPR Knock-in?
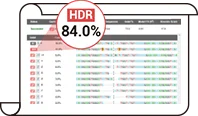
After years of R&D of Ubigene, based on the original EZ-editor™ gene-editing technique, Ubigene has upgraded it to EZ-HRex™ New Technique. With the new technology, the proportion of HDR genotypes can reach up to 84%. Get in touch with our experts now
- Efficiency: The efficiency of knock-in can be low, especially compared to knock-out techniques, because homology-directed repair (HDR) is less common than the alternative repair pathway, non-homologous end joining (NHEJ), which often leads to deletions or errors rather than precise insertions.
- Off-target Effects: Although CRISPR technology is highly specific, there is still the potential for off-target edits, where the CRISPR machinery cuts DNA at unintended sites, leading to unwanted genetic changes.
- Delivery: Delivering the CRISPR/Cas9 components (guide RNA, Cas9, and donor DNA) into cells, especially in vivo (in living organisms), is challenging. Efficient delivery systems are crucial for the success of CRISPR knock-in applications.
8. How to increase knock-In efficiency?
Ubigene has revolutionized gene editing with the new EZ-HRex™ technique, now enhanced with the innovative U+ Molecule. This breakthrough allows HDR genotype proportions to reach up to 84% at the post-transfection cell pool level. Additionally, Ubigene offers 4 tailored solutions to address diverse mutation requirements, ensuring maximum flexibility and precision in your experiments! Contact us now
9. How much does CRISPR Knock-in cost?
Ubigene offers global gene knock-in services, starting at just $6,480!