The H9C2 line of embryonic rat cardiomyocytes is a subclonal line of the original clonal cell line derived from embryonic BD1X rat heart tissue by Kimes and Brandt (1976). This cell line exhibits many of the properties of skeletal muscle and its parameters are particularly useful in preclinical tests of anticancer drugs, determining their cardiotoxicity, safety, and the possibility of moving to subsequent stages of clinical tests. Therefore H9C2 cell line is commonly used in numerous in vitro studies, some of which involve CRISPR/Cas9 technology to mediates the characteristics of H9C2 cells.
H9C2 cells retain some components of the signaling pathway essential for their differentiation into mature cardiac muscle cells. The cell line is used, in particular, for cardiotoxicity analyses of new, mainly anticancer drugs (e.g. doxorubicin), and studies on mechanisms of myocyte damage, and assessment of toxic effects of studied compounds on apoptosis and necrosis in cardiac myocytes. Embryonic H9C2 cardiomyocytes proliferate well in vitro conditions, allowing relatively easy culturing and are suitable to be a CRISPR gene knockout/knockin or over-expression model for in vitro studies of cardiac hypertrophy and supports current work with human cardiomyocyte cell lines for prospective molecular studies in heart development and disease.
Applications:
1. Effects of C3G Knockout on Proliferation and Apoptosis in H9C2 Cardiomyocytes
Previous studies found that C3G expression was significantly increased in the myocardium of the non-infarct area around the infarct in rats. Overexpressed C3G can promote cardiomyocyte survival and inhibit cytotoxicity while knocking down C3G can inhibit cardiomyocyte survival and increase cardiomyocyte apoptosis. The CRISPR/Cas9 system built-in knockout C3G recombinant lentivirus was used to infect H9C2 cardiomyocytes to study the speculated effect and mechanism of C3G knockout on the proliferation of H9C2 cardiomyocytes. H9C2 cardiomyocytes were infected with above lentiviruses respectively to investigate the effects of C3G [Crk SH3-domain-binding guanine nucleotide exchange factor] knockout on proliferation and apoptosis in H9C2 cardiomyocytes and their underlying mechanisms.
2. Using CRISPR-Cas9 gene-editing technology to knock out Tudor-SN gene of H9c2 cells to inhibit cell cycle arrest and proliferation
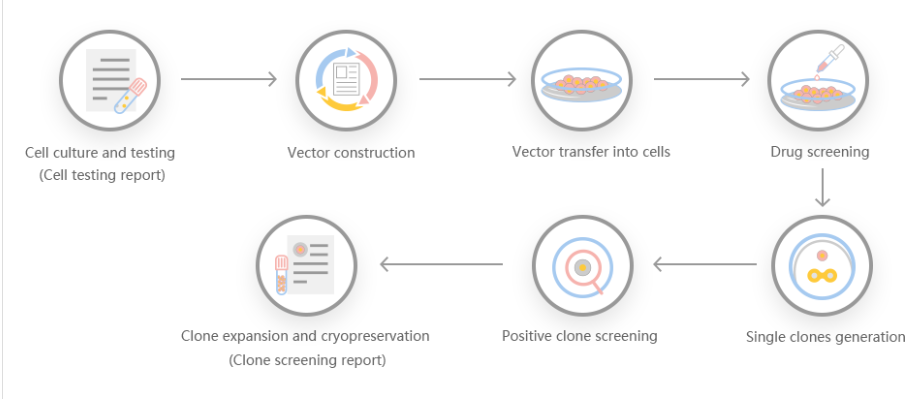
CRISPR-Cas9 gene-editing technology was used to knock out the Tudor-SN (Tudor staphylococcal nuclease) gene of rat myocardial H9c2 cells. Researchers observed its effect on H9c2 cell cycle and proliferation. The PX462 plasmid was selected as the vector, and the upstream and downstream sgRNA (single-guided RNA) that can specifically recognize the second exon of Tudor-SN gene in H9c2 cells were designed using software to construct a pair of recombinant plasmids. Subsequently, the pair of plasmids were co-transformed into H9c2 cells, and then positive monoclonal cells were selected for cultivation. Western blotting was used to identify the knockout effect, and the cell cycle and proliferation were detected by flow cytometry and CCK-8=experiment using the successfully knocked out cell lines. Western blotting results showed that Tudor-SN protein was not expressed in positive cells, and the Tudor-SN gene was successfully knocked out. Flow cytometry results showed that Tudor-SN gene knockout cells had a G1 phase arrest. The results of the CCK-8 experiment showed that the proliferation rate of KO cells slowed down. In this experiment, the Tudor-SN gene knockout cell line of H9c2 cells was successfully constructed, and the inhibition of Tudor-SN gene knockout on cell cycle arrest and proliferation was detected, which provided a study for the regulation of Tudor-SN gene on cardiomyocyte function. Convenient tools and research foundation.
3. Dock180 knockout inhibits proliferation and promotes apoptosis of rat derived H9C2 cardiomyocytes strain
To investigate the effects of dedicator of cytokinesis 1 (Dock180) knockout on proliferation and apoptosis in rat derived H9C2 cardiomyocytes and their mechanisms, a single guide RNA (sgRNA) targeting rat Dock180 gene was designed and constructed using CRISPR/Cas9 system. A plasmid contained above sgRNA was packaged into lentivirus and selected to knockout Dock180 in the cardiomyocytes. The result showed that Dock180 knockout with CRISPR/Cas9 H9C2 cells can inhibit proliferation and promote apoptosis via p-ERK1/2, Bcl-2, and Bax in H9C2 cardiomyocytes.
Gene knockout efficiency not only enables people to generate human glycosylation profiles and establish extended records, but also has many advantages. It is a particularly alternative recombinant protein expression system. First, followed by glutamic acid, the operation is simple, and recombinant proteins can be quickly produced by transient gene expression. Third, it can be used for stable recombinant protein production. Some researchers use gene knockout cutting efficiency systems to generate genes. Edit cell lines, recombine GLUL genomic loci to produce stable EPO transfected cell lines, and find that recombination contributes to stable expression of erythropoietin in humans.
According to the needs of customers, Well Biology combined with the target genes to design a stable gene knockout program.
Scheme 1: Small fragment gene knockout scheme. The gRNA is located in the intron at the border of exon 2. The number of exon coding sequences knocked out is not a multiple of three, which may result in backshifting after knocking out.
Scheme 2: Frame-shifting gene knockout scheme, where gRNA is set on the exon, the number of missing strings is not a multiple of three, and frame-shift mutation may occur after the knockout.
Scheme 3: Large fragment gene knockout scheme, the entire gene coding sequence is knocked out to obtain a large fragment knockout effect.
Ubigene Biosciences is co-founded by biological academics and elites from China, the United States, and France. We are located in Guangzhou Science City, which serves as a global center for high technology and innovation. Ubigene Biosciences has 1000㎡ office areas and laboratories, involving genome editing, cell biology technology, and zebrafish research. We provide products and services for plasmids, viruses, cells, and zebrafish. We aim to provide customers with better gene-editing tools for cell or animal research.
We developed CRISPR-U™ and CRISPR-B™(based on CRISPR/Cas9 technology) which is more efficient than general CRISPR/Cas9 in double-strand breaking, CRISPR-U™ and CRISPR-B™ can greatly improve the efficiency of homologous recombination, easily achieve knockout (KO), point mutation (PM) and knockin (KI) in vitro and in vivo.
References:
1. Nødvig CS, Hoof JB, Kogle ME, et al. Efficient oligonucleotide mediated CRISPR-Cas9 gene editing in Aspergilli. Fungal Genet Biol. 2018;115:78-89.
2. Nødvig CS, Nielsen JB, Kogle ME, Mortensen, UH. A CRISPR-Cas9 System for Genetic Engineering of Filamentous Fungi. PLoS One. 2015;10(7):e0133085. Published 2015 Jul 15.
3. Vanegas, K.G., Jarczynska, Z.D., Strucko, T. et al. Cpf1 enables fast and efficient genome editing in Aspergilli. Fungal Biol Biotechnol 6, 6 (2019).
4. Nayak T, Szewczyk E, Oakley CE, et al. A versatile and efficient gene-targeting system for Aspergillus nidulans. Genetics. 2006;172(3):1557-1566.
Genome Editing Platform
——Focusing on the Application of CRISPR-U™ and CRISPR-B™ Gene Editing Technology
Cell Biology Platform
——Focusing on primary cell
2. Provides culture strategies and related products for different cell types.3. Provides cell biology-related services such as cell isolation, extraction and validation.
 Subscribe Us
Subscribe Us Gene Editing Services
Gene Editing Services
 EZ-editor™
EZ-editor™ Red Cotton Gene knockout Project
Red Cotton Gene knockout Project








