Lignocellulosic biomass is mainly composed of cellulose, hemicellulose and lignin, and is the most abundant and renewable energy source on earth. The degradation of lignocellulosic biomass and the continuation of the carbon cycle in nature are mainly maintained by the action of microorganisms, including different fungal species such as Trichoderma, Aspergillus and Penicillium. Microorganisms play an important role in nature. The biomass-degrading enzymes produced by these organisms can also be used in various fields such as food, feed, papermaking and textile industries. Trichoderma reesei is a fungus and a well-known and efficient producer of cellulase and hemicellulase. Therefore, it is widely used by the enzyme industry to produce its own endogenous enzymes and produce heterogeneous proteins. Over the past two years, many studies have shown that the CRISPR/Cas9 system is a powerful genome editing method, and the knockout cell line can promote genetic changes in the genome of a variety of organisms. So far, there are no reports on the CRISPR/Cas9 system or other systems.
Although the technology has been successfully applied to yeast, the same is true for filamentous fungi and even genome editing methods in the model organism Neurospora crassa.Trichoderma reesei (Teelemorph Hypocrea jecorina) is a mesophilic soft saprophytic fungus, which is widely used in industry as a source of cellulase and hemicellulase for the hydrolysis of plant cell wall polysaccharides. Lignocellulosic biomass from crop residues, grass, wood and municipal solid waste represents a rich renewable resource, which is becoming more and more important as a future source of biofuels. Microorganisms are the liver that is harmless to the environment on earth. Although replacing gasoline with cellulosic ethanol can significantly reduce greenhouse gases in the atmosphere and reduce global warming, the high cost of hydrolyzing biomass polysaccharides into fermentable sugars is still a major obstacle that must be effectively overcome to make cellulosic ethanol effective Land commercialization. Since the cost of cellulase and hemicellulase greatly affects the price of bioethanol, much cheaper sources of these enzymes are needed. Therefore, genetic engineering techniques, gene knockout programs and DNA-mediated transformation systems have improved the T. reesei strains that produce industrial enzymes.
Efficient genome editing in Trichoderma reesei using the CRISPR/Cas9 system
The researchers demonstrated the establishment of the CRISPR/Cas9 system in the filamentous fungus Trichoderma reesei through specific codon optimization and in vitro RNA transcription. The results show that by inducing Cas9 expression, the CRISPR/Cas9 system is controllable and conditional. This system generates site-specific mutations in the target gene through effective homologous recombination, even using short homology arms. The system also provides a suitable and promising method for simultaneously targeting multiple genes. Our results indicate that the CRISPR/Cas9 system is the most powerful genome manipulation tool for Trichoderma reesei and other filamentous fungal species, which may accelerate the functional genomics and strain improvement studies of these filamentous fungi.
Rapid gene disruption in Trichoderma reesei using in vitro assembled Cas9/gRNA complex
In this study, the researchers tested two gene disruption methods in Trichoderma reesei using CRISPR/Cas9. The Cas9 expressed in the cell caused the accidental off-target gene destruction of Trichoderma reesei QM9414, which facilitated the insertion of 9 or 12 bp at 70 and 100 bp downstream of the target ura5. Therefore, by assembling Cas9 and gRNA in vitro, and then transforming the ribonucleoprotein complex and the plasmid containing the pyr4 marker gene into Trichoderma reesei TU-6, another method was established. When using gRNA targeting cbh1, it was found that 8 out of 27 transformants lost the ability to express CBH1, indicating that cbh1 was successfully destroyed by genome editing. Inserting large DNA fragments containing co-transformed plasmids, chromosomal genes, or these nucleotide mixtures into the disrupted cbh1 locus. Direct transformation of the Cas9/gRNA complex into cells is a fast method to disrupt the Trichoderma reesei gene and may be used in strains Improved and functional genomics research has been widely used.
Copper-controlled RNA interference system for reversible silencing of target genes in Trichoderma reesei
The researchers integrated the copper-responsive tcu1 promoter into the RNAi-mediated silencing system to develop a controllable RNAi-mediated silencing system, a Trichoderma reesei. As a proof of concept, when the respective RNAi fragments were knocked out, in the absence of copper, the prototrophic pyr4 gene was successfully knocked out. The high expression of cel7a and xyr1 genes induced by Avicel and knockout proved difficult to treat fab1 gene expression.
Ubigene Biosciences is co-founded by biological academics and elites from China, the United States, and France. We are located in Guangzhou Science City, which serves as a global center for high technology and innovation. Ubigene Biosciences has 1000㎡ office areas and laboratories, involving genome editing, cell biology technology, and zebrafish research. We provide products and services for plasmids, viruses, cells, and zebrafish. We aim to provide customers with better gene-editing tools for cell or animal research.
We developed CRISPR-U™ and CRISPR-B™(based on CRISPR/Cas9 technology) which is more efficient than general CRISPR/Cas9 in double-strand breaking, CRISPR-U™ and CRISPR-B™ can greatly improve the efficiency of homologous recombination, easily achieve knockout (KO), point mutation (PM) and knockin (KI) in vitro and in vivo.
Ubigene Biosciences is co-founded by biological academics and elites from China, the United States, and France. We are located in Guangzhou Science City, which serves as a global center for high technology and innovation. Ubigene Biosciences has 1000㎡ office areas and laboratories, involving genome editing, cell biology technology, and zebrafish research. We provide products and services for plasmids, viruses, cells, and zebrafish. We aim to provide customers with better gene-editing tools for cell or animal research.
We developed CRISPR-U™ and CRISPR-B™(based on CRISPR/Cas9 technology) which is more efficient than general CRISPR/Cas9 in double-strand breaking, CRISPR-U™ and CRISPR-B™ can greatly improve the efficiency of homologous recombination, easily achieve knockout (KO), point mutation (PM) and knockin (KI) in vitro and in vivo.
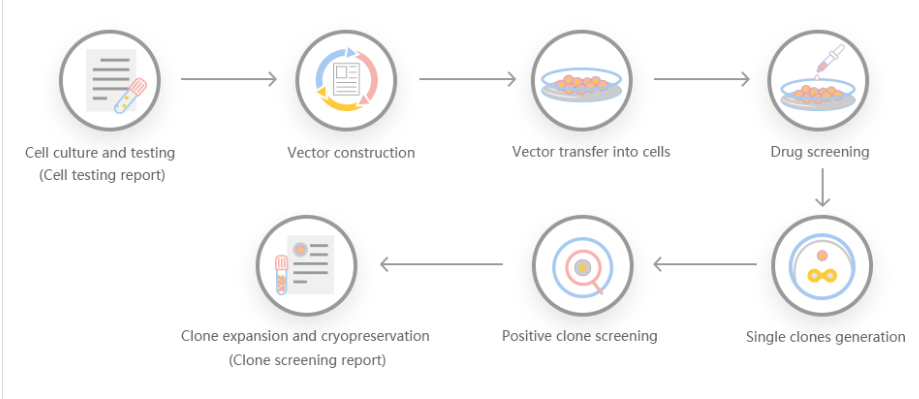
Genome Editing Platform
——Focusing on the Application of CRISPR-U™ and CRISPR-B™ Gene Editing Technology
1. Provides various types of gene-editing vectors for different species.
2. Provides different virus packaging services, including lentiviruses, adenoviruses and adeno-associated viruses.3. Provides high-quality services for gene knockout, point mutation and knockin cell lines.
Cell Biology Platform
——Focusing on primary cell
1. Provides over 400 types of primary cells.
2. Provides culture strategies and related products for different cell types.3. Provides cell biology-related services such as cell isolation, extraction and validation.
 Subscribe Us
Subscribe Us Gene Editing Services
Gene Editing Services
 EZ-editor™
EZ-editor™ Red Cotton Gene knockout Project
Red Cotton Gene knockout Project








