EGFR is a gene on chromosome 7p12, which encodes epidermal growth factor, a transmembrane glycoprotein of the protein kinase superfamily, and a receptor for members of the epidermal growth factor family. The binding of protein to ligand induces receptor dimerization and tyrosine autophosphorylation and leads to cell proliferation. Epidermal growth factor receptor (EGFR) has a basic role in normal physiology and cancer, making it a reasonable target for cancer treatment. Surprisingly, however, targeting has been proven to be classic, and inhibitors of ligand-stimulated EGFR signaling are largely ineffective in treating many EGFR-dependent cancers. Recent evidence has shown that both endogenous and treatment-induced cellular stress can trigger a robust and non-standard pathway for ligand-independent EGFR transport and signaling. knockout cell line can provide cancer cells with survival advantages and Resistance to treatment.
The CRISPR/Cas9 system targeting EGFR exon 17 eliminates NF-κB activation through epidermal genetic regulation of UBXN1 in UBwt/vIII glioma cells
Globally, glioblastoma (GBM) is the deadliest and most common intracranial tumor. Despite decades of research, the overall survival rate of GBM patients remains unchanged. Epidermal growth factor receptor (EGFR) amplification and gene mutations are considered to be negatively correlated with prognosis. In this study, the researchers used proteomics to determine that UBXN1 is a negative downstream regulator of EGFR mutation vIII (EGFRvIII). Through bioinformatics analysis, researchers found that UBXN1 is a factor that can improve the overall survival time of patients with glioma. The researchers also determined that in the presence of EGFRvIII, the down-regulation of UBXN1 is mediated by the up-regulation of H3K27me3. Because UBXN1 can negatively regulate NF-κB, researchers believe that EGFRwt/vIII activates NF-κB by inhibiting the expression of UBXN1. Importantly, the researchers used the latest genome editing tool CRISPR/Cas9 to knock out the EGFRwt/vIII on exon 17, and further proved that UBXN1 is negatively regulated by EGFRw t/vIII. In addition, EGFR/EGFRvIII knockout can benefit GBM in vitro and in vivo, which indicates that CRISPR/Cas9 is a promising treatment strategy for patients with EGFR amplification and EGFR mutations.
The transformation from non-small cell lung cancer to small cell lung cancer: molecular drivers and cells of origin
Lung cancer is the most common cause of cancer death worldwide. The two broad histological subtypes of lung cancer are small cell lung cancer (SCLC) and non-small cell lung cancer (NSCLC). Small cell lung cancer accounts for 15% of cases and non-small cell lung cancer accounts for 85% of cases, including adenocarcinoma. , Squamous cell carcinoma and large cell carcinoma. Although NSCLC and SCLC are generally regarded as different diseases due to their unique biological and genomic abnormalities, the view that these malignant diseases may share a common cell of origin has been supported. When resistance to EGFR tyrosine kinase inhibitors develops, part of the NSCLC with mutated EGFR returns to SCLC. This unexpected finding supports this idea. In addition, other case reports also described the coexistence of NSCLC and SCLC, further challenging the generally accepted view of their different ancestry. The researchers summarized the published clinical observations and biological basis, which combined SCLC and NSCLC histology, as well as the transformation of cancer from adenocarcinoma to SCLC. The researchers also discussed preclinical studies on common potential cells of origin, and speculated on how the genomics of each disease determines different differentiation pathways. origin
Stress-induced EGFR trafficking: mechanism, function, and therapeutic implications
Epidermal growth factor receptor (EGFR) has a basic role in normal physiology and cancer, making it a reasonable target for cancer treatment. Surprisingly, however, inhibitors that target classic, ligand-stimulated EGFR signaling have proven to be very ineffective in the treatment of many EGFR-dependent cancers. Recent evidence indicates that both endogenous and treatment-induced cellular stress can trigger a robust, non-standard pathway of ligand-independent EGFR transport and signaling, thereby providing cancer cells with survival advantages and resistance to treatment. The researchers reviewed the mechanism regulation of non-standard EGFR trading and signal transduction, as well as the pathological and therapeutic stresses that activate it. The researchers also discussed the significance of this pathway in the clinical treatment of EGFR overexpressing cancers.
Ubigene Biosciences was co-founded by biologists and elites from China, the United States and France. We are located in Guangzhou Science City, a global center of high technology and innovation. Ubigene Biosciences has 1,000 square meters of office area and laboratories, involving genome editing, cell biology technology and zebrafish research.
Reference
Huang K, Yang C, Wang QX, et al. The CRISPR/Cas9 system targeting EGFR exon 17 abrogates NF-κB activation via epigenetic modulation of UBXN1 in EGFRwt/vIII glioma cells. Cancer Lett. 2017;388:269-280. doi:10.1016/j.canlet.2016.12.011
Oser MG, Niederst MJ, Sequist LV, Engelman JA. Transformation from non-small-cell lung cancer to small-cell lung cancer: molecular drivers and cells of origin. Lancet Oncol. 2015;16(4):e165-e172. doi:10.1016/S1470-2045(14)71180-5
Tan X, Lambert P F, Rapraeger A C, et al. Stress-Induced EGFR Trafficking: Mechanisms, Functions, and Therapeutic Implications.[J]. Trends in Cell Biology, 2016, 26(5): 352-366.
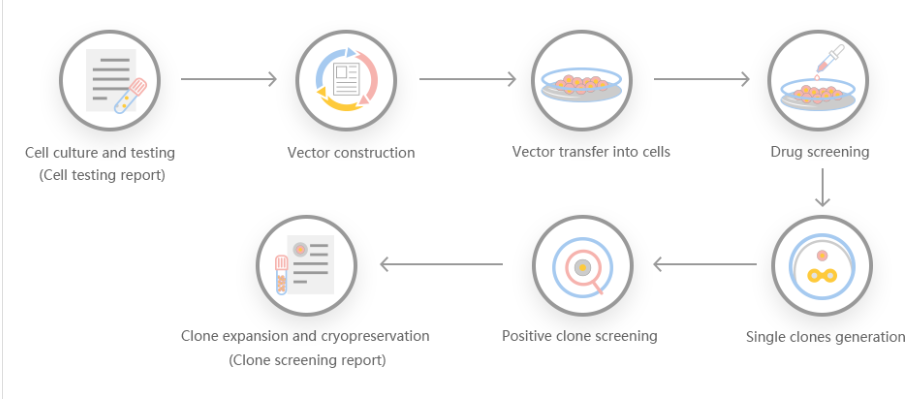
The efficiency of gene knock-out and cleavage can not only give people the ability to generate protein radical profiles and establish regulatory records, but also has many advantages, making it a particularly attractive recombinant protein expression system. First, it is carboxylated on glutamic acid and sulfated on tyrosine. Second, the operation is simple, and the recombinant protein can be quickly produced through transient gene expression. Third, it can be used for stable recombinant protein production. Some researchers used gene cell knockout and cutting efficiency systems to generate gene-edited cell lines, targeted sequencing of GLUL genomic loci, produced stable cell lines, and discovered the mechanism of stable expression of recombinant erythropoietin in humans .
According to customer needs, Yuanjing Biotechnology designs a stable gene transfer knockout program based on the target gene
Scheme 1: Small-segment gene knockout program, gRNA is set in the introns at both ends of exon 2, and the number of bases encoded by the knockout exon is not 3 times, and the knockout can cause frameshift.
Scheme 2: Frameshift gene knockout scheme, gRNA is set on the exon, the number of missing bases is not 3 times, and frameshift mutation can occur after knockout.
Scheme 3: Large-segment gene knockout scheme, knock out the coding sequence of the entire gene to achieve the effect of large-segment knockout.
Ubigene Biosciences is co-founded by biological academics and elites from China, the United States, and France. We are located in Guangzhou Science City, which serves as a global center for high technology and innovation. Ubigene Biosciences has 1000㎡ office areas and laboratories, involving genome editing, cell biology technology, and zebrafish research. We provide products and services for plasmids, viruses, cells, and zebrafish. We aim to provide customers with better gene-editing tools for cell or animal research.
We developed CRISPR-U™ and CRISPR-B™(based on CRISPR/Cas9 technology) which is more efficient than general CRISPR/Cas9 in double-strand breaking, CRISPR-U™ and CRISPR-B™ can greatly improve the efficiency of homologous recombination, easily achieve knockout (KO), point mutation (PM) and knockin (KI) in vitro and in vivo.
Genome Editing Platform
——Focusing on the Application of CRISPR-U™ and CRISPR-B™ Gene Editing Technology
1. Provides various types of gene-editing vectors for different species.
2. Provides different virus packaging services, including lentiviruses, adenoviruses and adeno-associated viruses.3. Provides high-quality services for gene knockout, point mutation and knockin cell lines.
Cell Biology Platform
——Focusing on primary cell
1. Provides over 400 types of primary cells.
2. Provides culture strategies and related products for different cell types.3. Provides cell biology-related services such as cell isolation, extraction and validation.
 Subscribe Us
Subscribe Us Gene Editing Services
Gene Editing Services
 EZ-editor™
EZ-editor™ Red Cotton Gene knockout Project
Red Cotton Gene knockout Project








