[Research highlight] A new idea of prevention and cure of COVID-19!
SARS-CoV-2 has caused a worldwide pandemic since 2019. Some studies have found that SARS-CoV-2 increases the number of autophagosomes, which seems to be a good breakthrough in prevention and treatment. However, a study on non-human primates showed that the FAD approved potential drug candidate chloroquine (CQ)/hydrochloroquine, as an autophagy regulator, could not inhibit SARS-CoV-2 infection, and it was found that it had no beneficial effect on the mortality of COVID-19 patients.
In view of this situation, Chao Shang, etc. (Changchun Veterinary Research Institute, Chinese Academy of Agriculture Sciences) believed that the mechanism of SARS-CoV-2 regulating autophagy in the host needs to be fully studied in different animal models. Recently, they published in the Journal of Virology (2020 IF: 5.1027) "Inhibition of autophagy suppresses SARS-CoV-2 replication and ameliorates pneumonia in hACE2 transgenic mice and xenografted human lung tissues" The results of SARS-CoV-2 infection on the established non-human primate Macaca fascilaris model and human angiotensin converting enzyme 2 (hACE2) transgenic mice, and constructs the original SARS-CoV-2 infection model based on human lung xenotransplantation mice have been shown in the article. It reveals the new mechanism of SARS-CoV-2 manipulating autophagosome formation, and found that autophagy inhibitors may be potential drugs for the treatment of SARS-CoV-2 virus infection, which provides a new idea for the study of the pathogenic mechanism of SARS-CoV-2 and its prevention and treatment.

SARS-CoV-2 infection was found to trigger autophagy in the Macaca fascularis infection model
Researchers analyzed autophagy by collecting SARS-CoV-2 infection in the lungs of the Macaca fascularis and the corresponding areas of the infected lung. It was found that the expression level of Atg5-Atg12 and LC3-I /LC3-I was significantly increased after SARS-CoV-2 infection (Autophagy activation).
There are two main ways for SARS-CoV-2 to increase autophagy
LC3-I, which is mainly located in the cytoplasm, is transformed into LC3-II after autophagy activation. LC3-II is integrated into the extended phagocyte membrane and controlled to extend into a complete autophagosome. Therefore, LC3-II can be used as a reliable autophagosome marker. Western blot (WB) showed that the expression level of LC3-II in Vero E6 and Huh-7 cell lines increased significantly after SARS-CoV-2 infection. In order to visually observe the accumulation of autophagosomes, researchers constructed Vero E6 and Huh-7 cell lines transformed with GFP-LC3 plasmid (constructed by Ubigene), and found that the level of GFP in cells increased significantly after SARS-CoV-2 infection for 24 hours (hpi), indicating that SARS-CoV-2 infection induced the formation and accumulation of autophagosomes. In addition, EGFP-mCherry-LC3 transient Vero E6 cell line (constructed by Ubigene) was constructed to visualize the transfer of LC3 from autophagosome to lysosome. It was found that SARS-CoV-2 increased the number of autophagosomes in two ways: (1) the biogenic events of autophagosome increased; (2) the fusion of autophagosome and lysosome decreased.
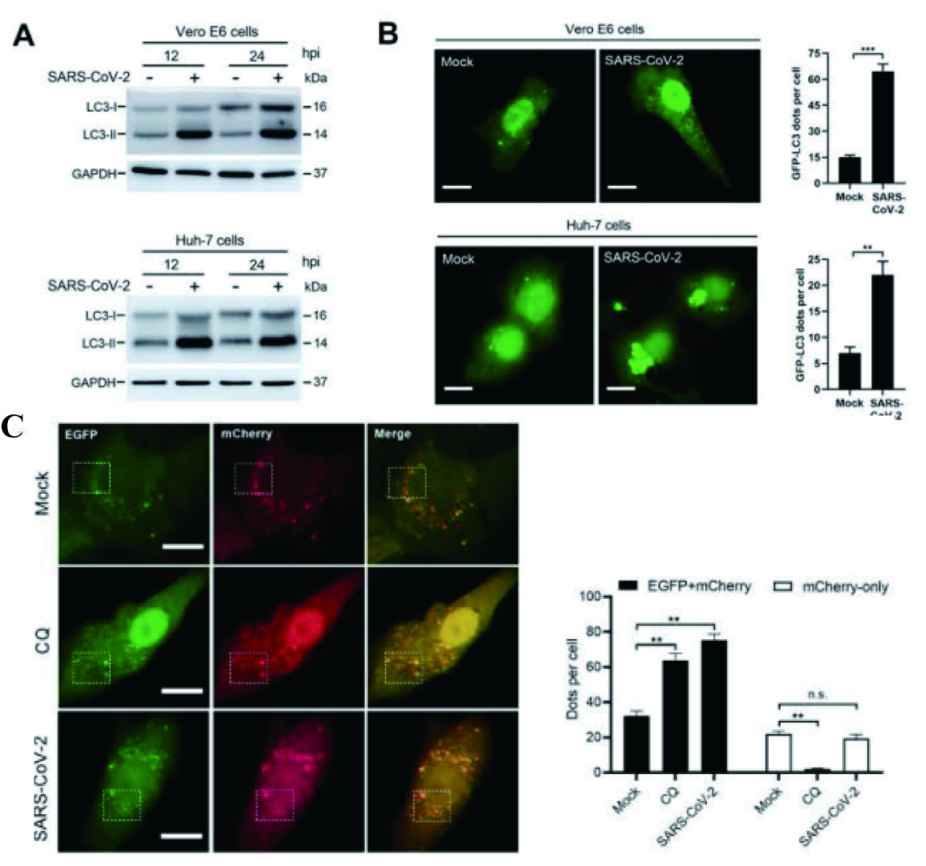
Figure 1 SARS-CoV-2 induces autophagosome formation in cell lines and blocks autophagosome-lysosome fusion
Autophagy related cascade reactions were analyzed to determine the relationship between autophagosome formation and virus replication
In order to further clarify the mechanism of SARS-CoV-2-induced autophagosome formation, researchers analyzed a series of signal cascades involved in autophagosome formation in the Vero E6 cell line infected with SARS-CoV-2. mTORC1 signaling pathway is the most common upstream autophagy inhibitory regulator. Researchers choose to monitor its activity by phosphorylation of its downstream substrate. It was found that SARS-CoV-2 not only inhibited Akt-mTOR signaling pathway, but also activated ULK-1-Atg13 and VPS34-VPS15-Beclin1 complexes. Moreover, in order to determine the relationship between autophagosome formation and virus replication and the key factors of virus replication, researchers used broad-spectrum autophagy inhibitor 3-MA and VPS34 inhibitor SAR405. It was found that the pretreatment of 3-MA and SAR405 significantly reduced the virus load in the Vero E6 cell line, indicating the importance of autophagy formation and VPS34 complex activation for SARS-CoV-2 virus replication.
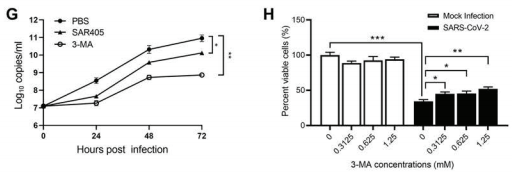
Figure 2 Autophagy formation and VPS34 complex activation promote the replication of the SARS-CoV-2 virus
Inhibition of autophagy can reduce SARS-CoV-2 replication and improve pneumonia in mice
Researchers used two different animal models to simulate the infection process of the SARS-CoV-2 in vivo. In the hACE2 transgenic mouse model, inhibition of autophagy can inhibit the replication of SARS-CoV-2 virus and improve pneumonia. On the contrary, rapamycin induced autophagy can promote viral autophagy and aggravate the pathological process of pneumonia.
In the innovative mouse model (transplanted with human lung tissue), they proved that SARS-CoV-2 could replicate itself in human lung tissue and spontaneously produce pneumonia.
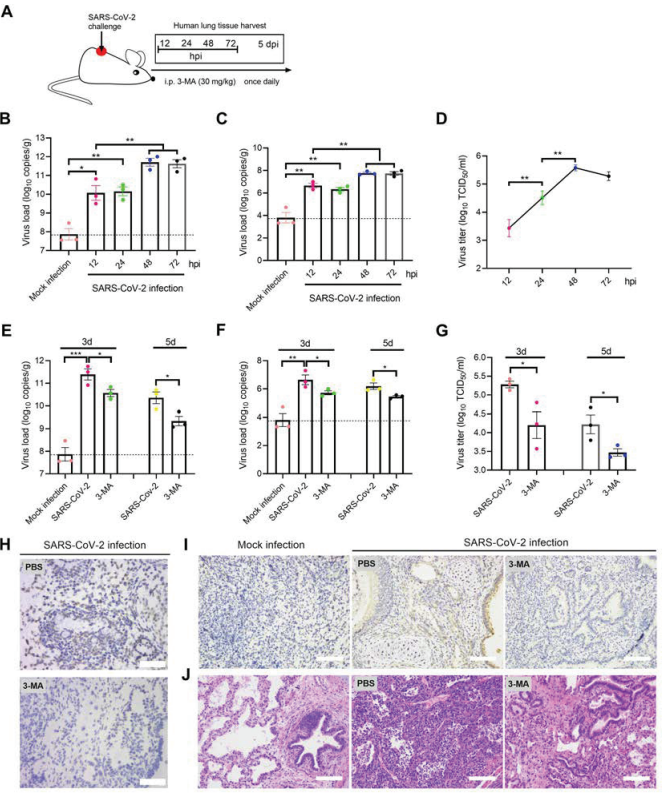
Figure 3 Inhibition of autophagy can inhibit SARS-CoV-2 replication and improve pneumonia in the mouse model (transplanted with human lung tissue)
In conclusion, researchers confirmed that autophagy promotes the replication of SARS-CoV-2 and promotes the process of pneumonia, and proposed that autophagy inhibitors may be potential drugs for the treatment of SARS-CoV-2 virus infection. Through the mutual verification of in vivo and in vitro experiments. This study lays a theoretical foundation for the in-depth study of virus cell interaction in the future, and provides a useful idea for the development of autophagy related antiviral drugs.
Ubigene focuses on cell line engineering. In this study, engineered Vero E6 and Huh-7 cell lines were provided to explore the relationship between SARS-CoV-2 virus replication and autophagy induction mechanism.
Ubigene specializes in gene-editing cell line, stable cell line generation, virus packaging, etc. Ubigene has about 2000 in-stock KO cell lines in our exclusive KO cell line bank, and EZ-editor™ Gene-editing Series Products, such as Gene Knockout kit help to achieve successful gene-editing! For more information about our services, please inquire >>
 Subscribe Us
Subscribe Us Gene Editing Services
Gene Editing Services
 EZ-editor™
EZ-editor™ Red Cotton Gene knockout Project
Red Cotton Gene knockout Project












