ZNF432 knockout U2OS cell line helped discover drug resistance related factors of ovarian cancer


ZNF432 knockout U2OS cell line helped discover drug resistance related factors of ovarian cancer

Recently, ZNF protein was confirmed as a key factor regulating the genome integrity of mammalian cells. In order to explore the possibility that ZFP can be used as an effector of DNA repair based on homologous recombination (HR), Jean Yves Masson team of Laval University used the ZNF432 knockout U2OS cell line constructed by Ubigene to carry out a series of experiments, and found that ZNF432 deletion in cancer cells would accelerate DNA repair, lead to the weakening of PARPi effect, and make ovarian cancer cells develop drug resistance, confirming that ZNF432 is a new HR inhibitor, which successfully broadened the new way to study the efficacy of PARPi [1]. The article entitled ZNF432 stimulates PARylation and inhibits DNA resection to balance PARPi sensitivity and resistance was published on Nucleic Acids Research(IF=14.9).
Poly(ADP-ribosy)lation (PARylation) modification has been confirmed to be coordinately involved in DNA damage response and is associated with DNA repair. In order to find PAR-related genes that affect DNA double strand break (DSB) repair, the authors selected seven ZFPs identified as PAR-binding proteins to evaluate their potential role in homologous recombination repair (HR). In the pre-screen experiment, siRNA-induced gene silencing for each ZFP target was performed and ZNF432 showed the most significant effect among them (Figure 1). After that, the authors used the ZNF432 knockout U2OS cell line developed by Ubigene CRISPR-U™ technology and revealed that the expression of RAD51 and BrdU proteins involved in DNA repair was increased, which significantly increased the accumulation of pRPA, confirming that ZNF432 is a new DDR factor and contributes to HR-mediated DSB repair.
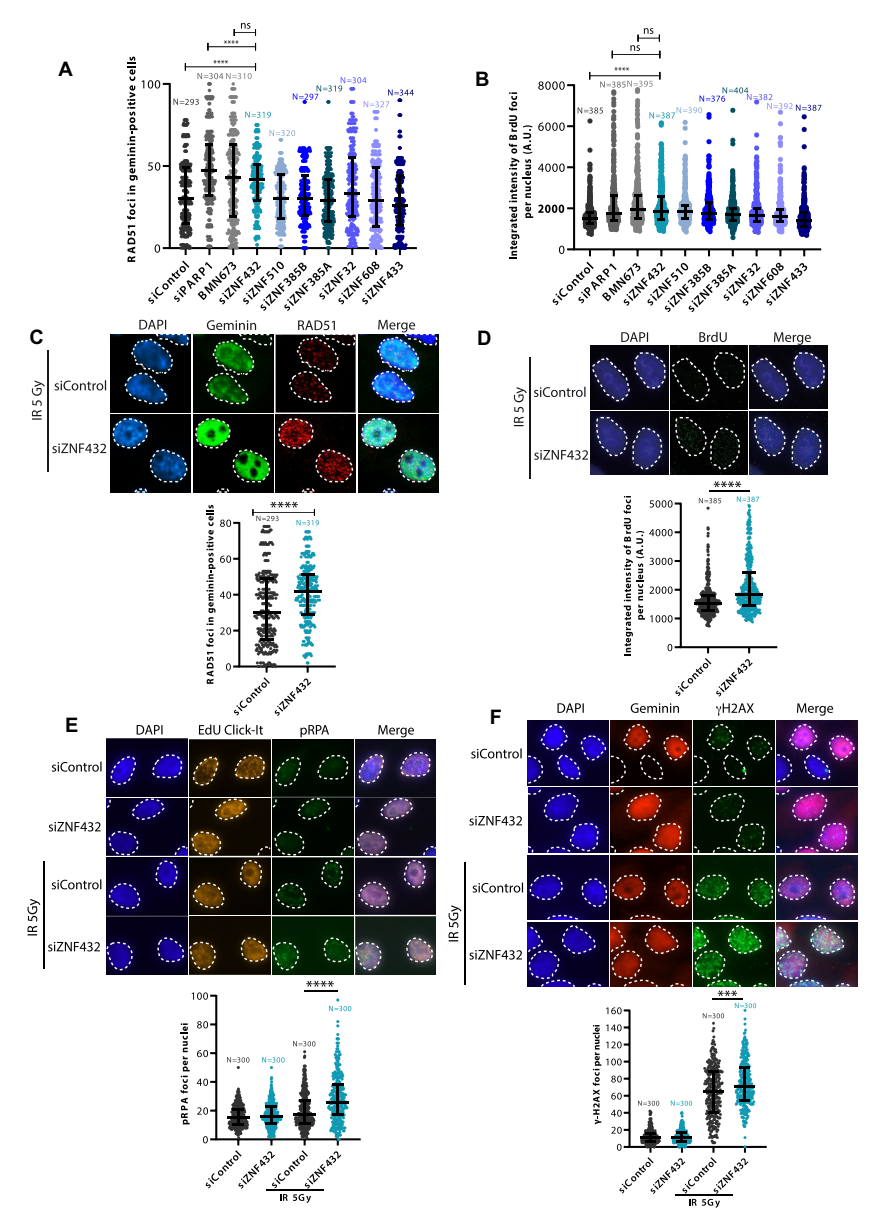
Figure 1. Analysis and identification of potential DDR regulators in ZFPs
ZNF432 has a KRAB domain and a zinc finger (ZNF) domain consisting of 16 repeats. Currently, five serine PARylation sites have been identified, three of which are located in the ZNF domain (Figure 2). KRAB targets proteins in the nucleoplasm through interacting with TRIM28/KAP1, while ZNFs uniformly targets proteins in the whole nucleus. The cooperative activity of KAP1-KRAB-ZNFs can make KRAB-ZFPs undergo precise nucleoplasmic localization, and it proves that ZNF432 will relocate to the damaged site and participate in the DNA repair process. GFP fusion proteins of KRAB and ZNFs were expressed in 293T cells and monitored using micro-IR. Both domains were expressed as nuclear proteins. The enrichment dynamics of GFP-ZNF432 under normal conditions were compared with those under PARP inhibition (PARPi) with BMN673 and in PARP-1 knockout cells. The localization of GFP-ZNF432 in laser-induced DSBs was found to be dependent on PAR.
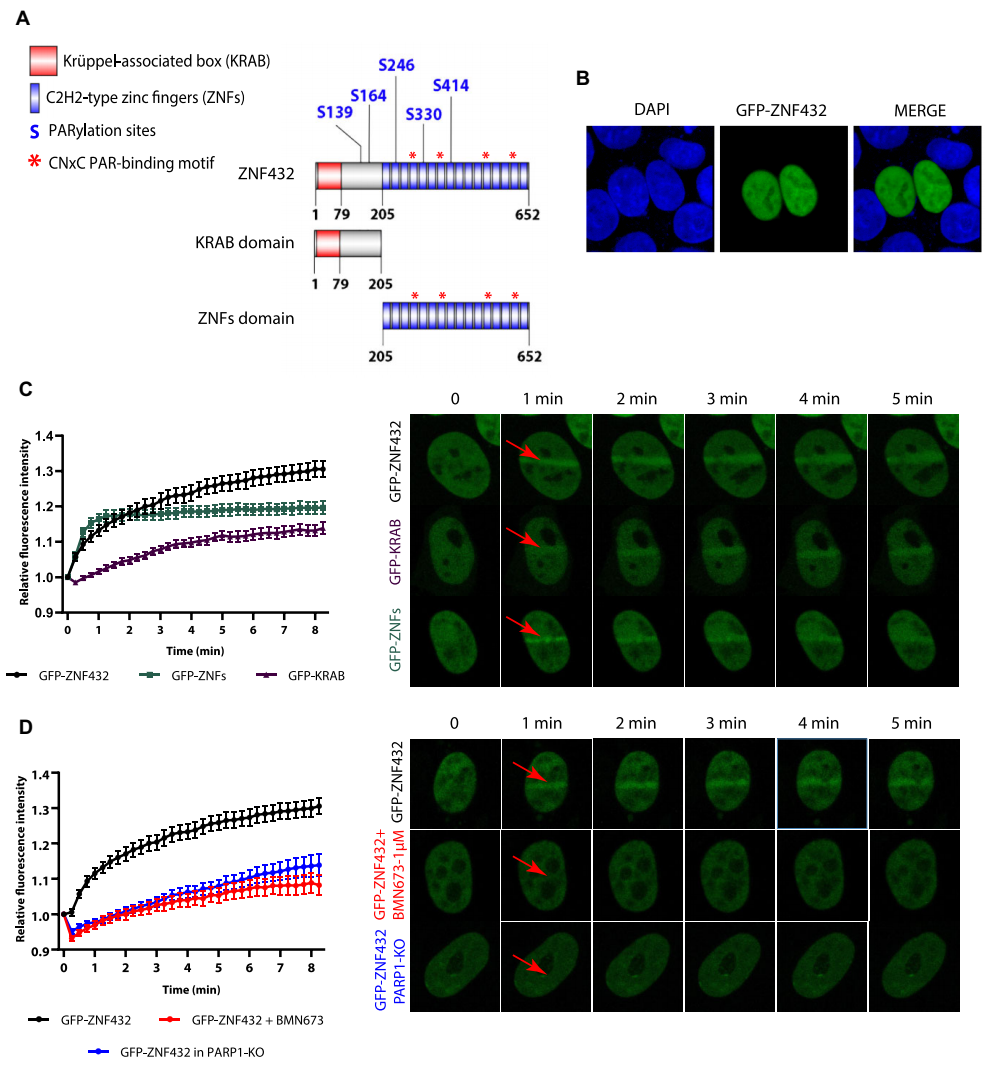
Figure 2. Inhibition of DNA repair by ZNF432 depends on the synthesis of PARP-1 and PAR
DNA damage response (DDR) proteins to DNA damage usually only rely on PAR-induced chromatin remodeling. To further discuss the response of PAR to ZNF432 DNA damage sites, laser-induced experiments revealed that ZNF432 binds PAR in vitro (Figure 3). KRAB-related protein was confirmed to be one of the proteins interacting with ZNF432 in HEK293T cells, indicating a robust promiscuous interaction between proteins. Using PARP-1-depleted cells, it was observed that the KAP1-ZNF432 protein complex was very stable and did not dissociate in the absence of PARP-1 nor after PARP-1 activation. After PARP-1 was activated, hundreds of proteins were identified in the ZNF432 protein interaction network. These results indicate that the assembly of ZNF432-interacting proteins can be remodeled when induced by PARP-1, further supporting that ZNF432 regulates DDR through a PARP-1-dependent pathway. Using ZNF432 KO cells, a decrease in PARylation expression was observed compared with overexpressing cells, further validating that PARP-1 activation is one of the most critical steps in DNA damage signaling and repair.
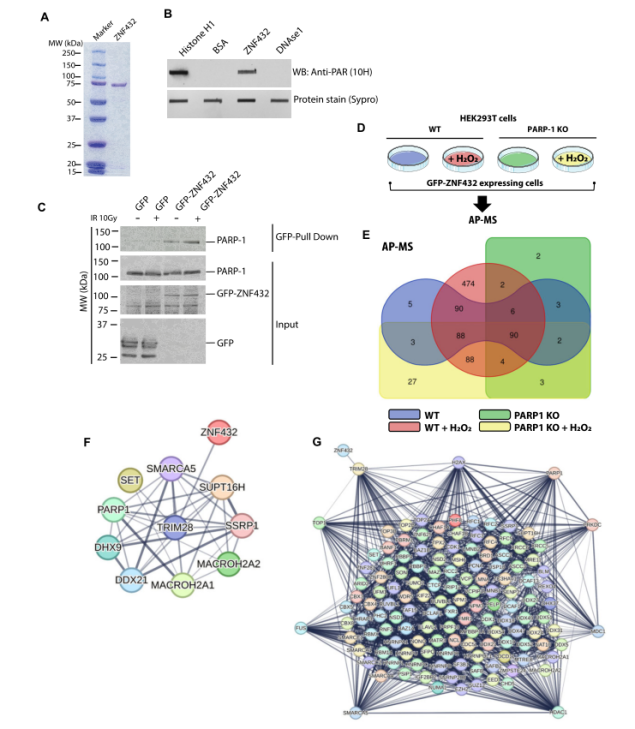
Figure 3. Interaction of ZNF432 with PAR and PARP-1
Because HR-deficient tumor cells are highly sensitive to PARPi, the authors evaluated whether it would lead to PARPi resistance by restoring HR in these cells through ZNF432 depletion. First, they observed that the attenuation of ZNF432 expression promoted the resistance of U2OS cells to PARPi (Figure 4), and subsequently restored the sensitivity of PARPi through the complementation experiment.
Gene expression analysis of ovarian cancer tissue samples showed that the overall expression level of ZNF432 in ovarian cancer samples was lower than that in normal ovarian samples. Compared with the healthy population, the expression of ZNF432 in endometroid ovarian cancer patients was significantly reduced. Analysis of more samples showed that regulating the expression of ZNF432 in ovarian cancer cells could sensitize ovarian cancer cells to PARPi. Ovarian cancer cell line COV362 and drug-resistant version COV362-R were selected to further evaluate the effect of ZNF432 overexpression in high-grade serous ovarian cancer cell models. It was found that ZNF432 overexpression made COV362 and COV362-R cells sensitive to drugs, and knockdown would promote resistance to drugs.
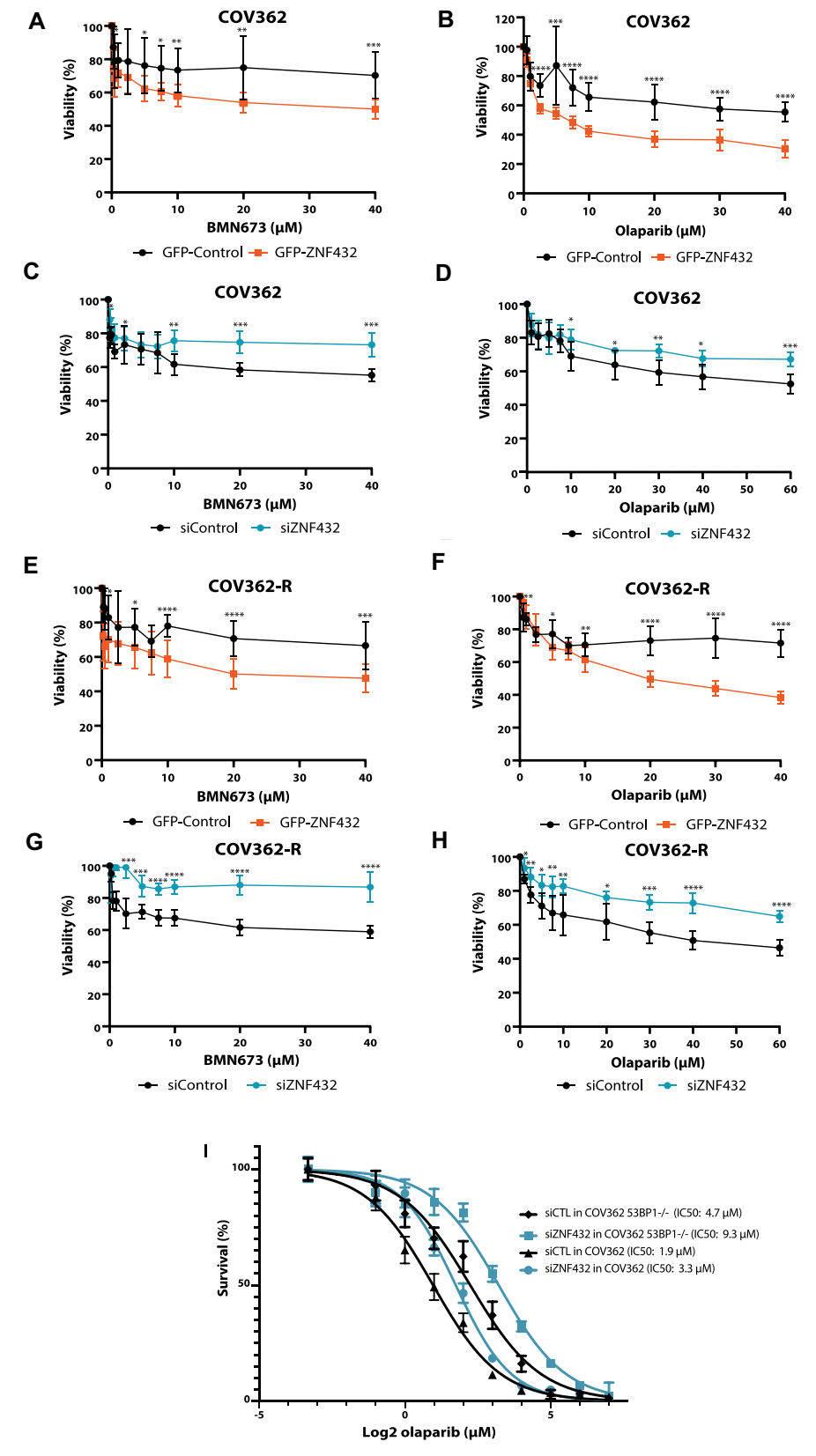
Figure 4. Regulation of ZNF432 expression affects drug resistance of ovarian cancer cells
In this study, the authors reported a new role of ZNF432, a ZNF protein, as a PAR reader to regulate DNA damage response. The deletion of ZNF432 in the cell system will lead to resistance to PARP inhibitors. In conclusion, the authors' findings support an emerging concept that ZNF-containing proteins can regulate the phosphorylation of PARPi. ZNF432 is a new HR inhibitor that participates in the cellular response to DNA damage and PARPi. This study provides important value for the prognosis of ovarian cancer.
Reference:
[1] O’Sullivan J, Kothari C, Caron M C, et al. ZNF432 stimulates PARylation and inhibits DNA resection to balance PARPi sensitivity and resistance[J]. Nucleic Acids Research, 2023, 51(20): 11056-11079.


