Introduction:
Aspergillus fumigatus is a species of fungus in the genus Aspergillus and is one of the most common Aspergillus species to cause disease in individuals with immunodeficiency. It can be found throughout the environment, including in soil, plant matter, and household dust. Humans and animals constantly inhale numerous conidia of this fungus. The conidia are normally eliminated in the immunocompetent host by innate immune mechanisms, and aspergilloma and allergic bronchopulmonary aspergillosis, uncommon clinical syndromes. Thus, A. fumigatus was considered for years to be a weak pathogen. Nowadays, Aspergillus fumigatus becomes more and more popular in research, because this specie plays an important role in the environment and human life. Also, CRISPR/Cas9 is a novel genome-editing system that has been successfully established in Aspergillus fumigatus. Scientists have generated a CRISPR system for Aspergillus fumigatus to study its function by knockout cell lines, gene knockin, point mutation, etc.
Aspergillus fumigatus is one of the most common causes of aspergillosis. The first one is allergic bronchopulmonary aspergillosis, it is an allergic reaction to the Aspergillus spores. This reaction can lead to damage in your airways and lungs. It is often found in people that have conditions such as asthma and cystic fibrosis. The second one is Chronic pulmonary aspergillosis, which can occur in people with chronic lung conditions that cause air spaces called cavities to form in the lung. The third one is invasive aspergillosis, it is the most severe form of aspergillosis and can be fatal if not treated. It occurs when an aspergillosis infection begins in the lungs and spreads to other parts of the human body, such as skin, brain, or kidney. Invasive aspergillosis occurs only in people who have a severely weakened immune system.
Development of the CRISPR/Cas9 System for Targeted Gene Disruption in Aspergillus fumigatus
To test the CRISPR/Cas9 systems feasibility for targeted gene disruption in A. fumigatus. As a proof of principle, researchers first demonstrated that CRISPR/Cas9 can indeed be used for high-efficiency (25 to 53%) targeting of the A. fumigatus polyketide synthase gene (pksP), as evidenced by the generation of colorless (albino) mutants harboring the expected genomic alteration. Researchers further demonstrated that the constitutive expression of the Cas9 nuclease by itself is not deleterious to A. fumigatus growth or virulence, thus making the CRISPR system compatible with studies involved in pathogenesis. Taken together, these data demonstrate that CRISPR can be utilized for loss-of-function studies in A. fumigatus and has the potential to bolster the genetic toolbox for this important pathogen.
Using CRISPR to Gene Manipulation in Aspergillus fumigatus
The current state of the technology relies heavily on DNA-based expression cassettes for delivering Cas9 and the guide RNA (gRNA) to the cell. Therefore, the power of technology is limited to strains that are engineered to express Cas9 and gRNA. To overcome such limitations, researchers developed a simple and universal CRISPR-Cas9 system for gene deletion that works across different genetic backgrounds of A. fumigatus. The system employs in vitro assembly of dual Cas9 ribonucleoproteins (RNPs) for targeted gene deletion. Additionally, the CRISPR-Cas9 system utilizes 35 to 50 bp of flanking regions for mediating homologous recombination at Cas9 double-strand breaks (DSBs). Similar deletion efficiencies were obtained in the clinical isolate DI15-102. The data shows that in vitro-assembled Cas9 RNPs coupled with microhomology repair templates are an efficient and universal system for gene manipulation in A. fumigatus. In this study, tackling the multifactorial nature of virulence and antifungal drug resistance in A. fumigatus requires the mechanistic interrogation of a multitude of genes, sometimes across multiple genetic backgrounds. Classical fungal gene replacement systems can be laborious and time-consuming and, in wild-type isolates, are impeded by low rates of homologous recombination. The simple and universal CRISPR-Cas9 system for gene manipulation in this study generates efficient gene targeting across different genetic backgrounds of A. fumigatus.
Ubigene developed CRISPR-B™ which optimizes the microbial gene-editing vectors and process. The efficiency and accuracy are much higher than traditional methods. CRISPR-B™ can be used in gene editing of bacteria and fungi. Easily achieve microbial gene knockout (KO), point mutation (PM), and knockin (KI).
The efficiency of gene knock-out and cleavage not only allows people to have the ability to generate protein generative profiles and establish extended records, but also has many advantages. It is a special alternative recombinant protein expression system. Sulfate on tyrosine. Second, the operation is simple, and the recombinant protein can be quickly produced through transient gene expression. Third, it can be used for stable recombinant protein production. Some researchers use gene cell knockout cutting efficiency system to generate genes. Edited the cell line, recombined the GLUL genome site, produced a stable transgenic cell line of EPO, and discovered the mechanism of recombination to promote the stable expression of erythropoietin in the human body.
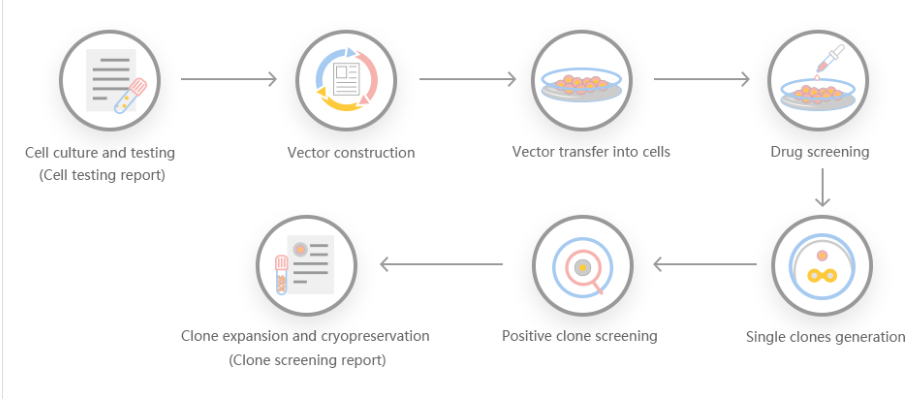
According to customer needs, Yuanjing Biotechnology designs a stable gene transfer knockout program based on the target gene.
Scheme 1: Small-segment gene knockout program, gRNA is set in the intron at the boundary of exon 2, and the number of coding sequences of knockout is not 3 times, and the knockout can cause frame shift.
Scheme 2: Frameshift gene knockout scheme, gRNA is set on the exon, the number of missing strings is not 3 times, and frameshift mutations can occur after knockout.
Scheme 3: Large-segment gene knockout scheme, knock out the coding sequence of the entire gene to achieve the effect of large-segment knockout.
Ubigene Biosciences is co-founded by biological academics and elites from China, the United States, and France. We are located in Guangzhou Science City, which serves as a global center for high technology and innovation. Ubigene Biosciences has 1000㎡ office areas and laboratories, involving genome editing, cell biology technology, and zebrafish research. We provide products and services for plasmids, viruses, cells, and zebrafish. We aim to provide customers with better gene-editing tools for cell or animal research.
We developed CRISPR-U™ and CRISPR-B™(based on CRISPR/Cas9 technology) which is more efficient than general CRISPR/Cas9 in double-strand breaking, CRISPR-U™ and CRISPR-B™ can greatly improve the efficiency of homologous recombination, easily achieve knockout (KO), point mutation (PM) and knockin (KI) in vitro and in vivo.
References:
1. GAN Shi-Hu, CUI Xiao-Teng, MA Jin-Zheng, FANG Li-Jiao, LIU Ming-Xia, REN Yuan-Yuan, CAO Xiao-Na, YANG Jie, SU Chao. 2.Using CRISPR-Cas9 gene-editing technology to knock out the Tudor-SN gene of H9c2 cells to inhibit cell cycle arrest and proliferation. Chinese Journal of Biochemistry and Molecular Biology, 2018.07.10.
2. Deng Qin, Liu Cheng, Zhang Jing, Li Gang. The effect of knocking out C3G on the proliferation and apoptosis of H9C2 cardiomyocytes. Chinese Journal of Cell Biology: 1-6[2020-06-09].
3. HU Su-lei, LI Gang, FU Yan-bo, DENG Qin, LIU Cheng. Dock180 knockout inhibits proliferation and promotes apoptosis of rat derived H9C2 cardiomyocytes strain. Basic & Clinical Medicine. 2017, 37.
Genome Editing Platform
——Focusing on the Application of CRISPR-U™ and CRISPR-B™ Gene Editing Technology
2. Provides different virus packaging services, including lentiviruses, adenoviruses and adeno-associated viruses. 3. Provides high-quality services for gene knockout, point mutation andknockin cell lines.
Cell Biology Platform
——Focusing on primary cell
2. Provides culture strategies and related products for different cell types.3. Provides cell biology-related services such as cell isolation, extraction and validation.
 Subscribe Us
Subscribe Us Gene Editing Services
Gene Editing Services
 EZ-editor™
EZ-editor™ Red Cotton Gene knockout Project
Red Cotton Gene knockout Project








