New insights into osteoporosis: GSTP1 mediated S-glutathionylation of Pik3r1 regulates osteoclast formation


New insights into osteoporosis: GSTP1 mediated S-glutathionylation of Pik3r1 regulates osteoclast formation
Osteoporosis is an age-related metabolic disease that has a direct relationship with bone density, and the therapeutic potential of GSTP1 (Glutathione S-transferase P1) in a variety of diseases has been uncovered. The study found GSTP1 may have some association with bone mineral density (BMD) through genetic association by Mendelian randomization and single-cell sequencing analysis. To explore the specific mechanism underlying the effect of GSTP1 on BMD, the researcher through key experiments, such as redox mass spectrometry, Co immunoprecipitation, non-denaturing and non-reducing immunoblotting, flow cytometry, mass spectrometry analysis, and CRISPR/Cas9 gene editing, found that GSTP1 can upregulate S-glutathione phosphorylation at Cys498 and Cys670 of Pik3r1, thereby inhibiting its phosphorylation, which in turn affects osteoclast differentiation in vitro by modulating changes in autophagic flux via the Pik3r1-AKT-mTOR axis, and finally regulated osteoclast formation in vitro.
Shigui Yan, et al, from Orthopedics Research Institute of Zhejiang University published an article in REDOX BIOLOGY, with the title “GSTP1-mediated S-glutathionylation of Pik3r1 is a redox hub that inhibits osteoclastogenesis through regulating autophagic flux”(IF2022:10.787).

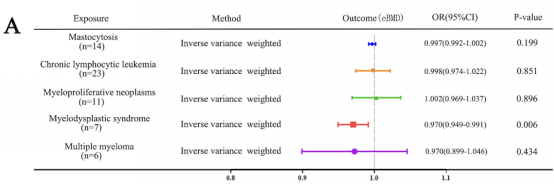
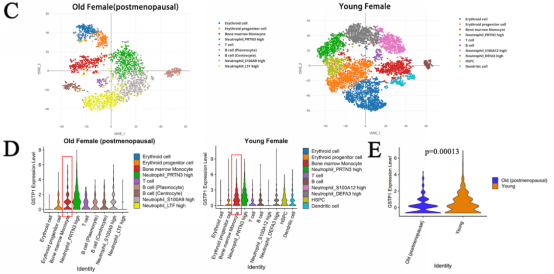
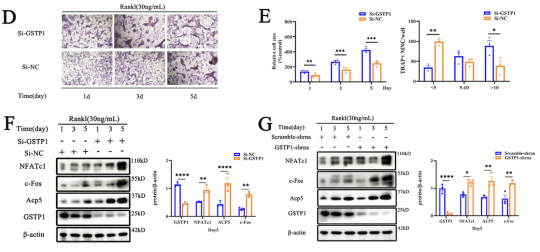
The study was first performed by Mendelian randomization analysis to demonstrate a potential association between MDS and bone mineral density (BMD). Summarized the signal transduction proteins and possible targets of MDS that have been reported, it was found that GSTP1 may affect BMD. Using RT-qPCR to measure the expression of RUNX2 (an osteoblast-specific gene) and knockdown or overexpression of GSTP1, the researcher found that GSTP1 has little relationship with osteoblast differentiation. Also by a combination of bone marrow single-cell RNA-sequencing and Western blot, the results showed that GSTP1 is highly expressed in bone marrow monocytes and differentially expressed between older and younger women, so the effect of GSTP1 on BMD was found to occur more likely through osteoclasts.

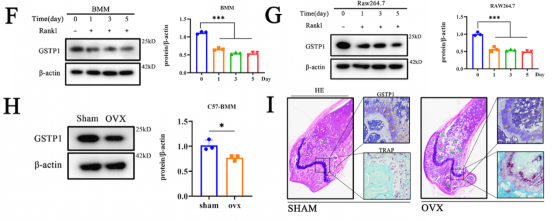
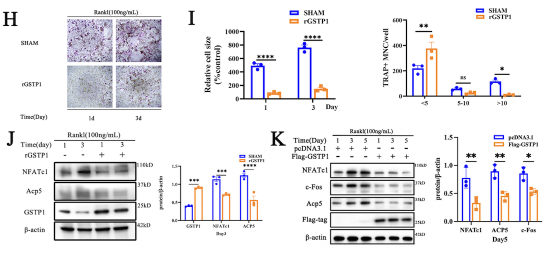
Autophagy has a wide range of effects on osteoclast differentiation, and in this article, the researcher continued to explore how GSTP1 affects osteoclasts from the perspective of autophagy. The cells presented more autophagic vesicles and phagocytic vesicles when the cells were stimulated with rGSTP1. The mammalian autophagy protein LC3 was selected as a marker of autophagosomes, and the researcher detected the altered autophagic flux during osteoclastogenesis in different cell lines upon overexpression or knockdown of GSTP1 by ratiometric flow cytometry and autophagy double labeled lentivirus. Whereas rapamycin, an autophagy activator, reversed the osteoclastogenesis promoted by low-level GSTP1 by activating autophagy flux. The above results demonstrated that GSTP1 could regulate osteoclastogenesis by regulating autophagic flux during osteoclast differentiation.
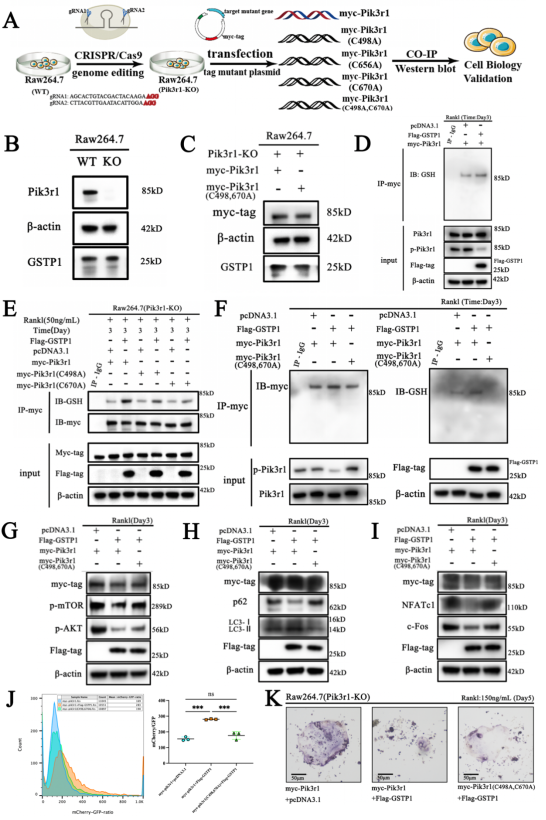
Rapamycin targets mTOR, which is involved in mammalian autophagy mainly through ERK or PI3K-AKT pathway activation. To determine whether the regulation of autophagy by GSTP1 during osteoclast differentiation is accomplished through mTOR, BMMs were differentiated for 5 days under stimulation, and the phosphorylation levels were not different from those of the control group. So, knockdown and transfection were ultilized to explore the relationship between the levels of GSTP1 and phosphorylated Pik3r1, phosphorylated AKT levels, and the results suggest that the autophagic flux regulated by GSTP1 is accomplished through the Pik3r1-AKT-mTOR signaling cascade.

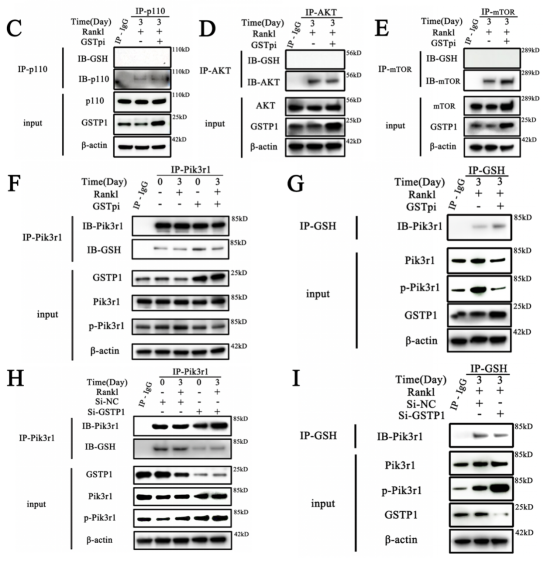
To further verify whether the regulation of the kinase activity downstream of GSTP1 is related to S-glutathione, the researcher explored the more microscopic protein molecular structure of PIK3R1. First, preliminary sequence alignment of different species revealed that four cysteines (Cys498, Cys656, Cys659, Cys670) in Pik3r1 were evolutionarily conserved, suggesting that cysteine residues play important roles in the regulation of Pik3r1 function. Co-immunoprecipitation results showed that overexpression and knockdown of GSTP1 altered the S-glutathionylation level of Pik3r1 during osteoclastogenesis, which in turn downregulated the phosphorylation level of Pik3r1 as a kinase.
Next, S-glutathionylation could take place at the Cys498 and Cys670, demonstrated by non-reducing and non-denaturing co-immunoprecipitation as well as by redox associated modification mass spectrometry. In addition, the results of molecular protein docking also showed that Cys498 and Cys670 were more prone to interact with GSH. The 2D images clearly showed that upon covalent docking of glutathione with the protein Pik3r1-CYS498, glutathione forms hydrogen bonds with GLN499 and GLU502 and electrostatic interactions with LYS532 and GLU502 occurred; Likewise, 2D pattern of the docking of Pik3r1-CYS670 and the ligand GSH revealed that the small molecule GSH formed hydrogen bonds with HIS669, HIS668, ARG649, and VAL671 and electrostatic interactions with ARG649.
Taken together, the researcher identified Cys498 and Cys670 as two cysteine residue sites for S-glutathionylation of Pik3r1.



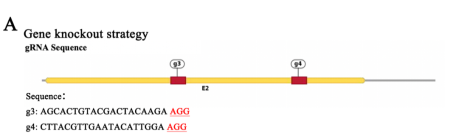
To elucidate the role of GSTP1-mediated S-glutathionylation of Pik3r1 in osteoclastogenesis, the researcher first knock out Pik3r1 in RAW264.7 cells using CRISPR/Cas9 gene-editing technology to exclude the influence of endogenous Pik3r1 and then generated three single-site mutation plasmids and one double-site mutation plasmid by replacing the cysteine site where S-glutathionylation may occur with alanine. Finally, these mutant plasmids and the wild-type myc-Pik3r1 plasmid were transfected into Pik3r1-KO RAW264.7 cells for subsequent experiments. Ubigene successfully constructed and provided the Pik3r1-KO RAW264.7 using CRISPR/Cas9 gene-editing technology (sees below figures), which laid an important cornerstone for subsequent mutation complementation validation.
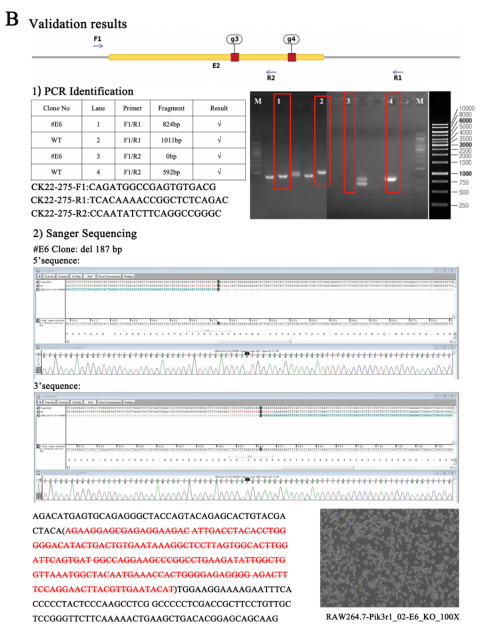
GSTP1-mediated S-glutathionylation of PIK3R1 regulates osteoclastogenesis
During osteoclastogenesis, the results of Co-immunoprecipitation showed that the level of GSTP1-mediated Pik3r1 S-glutathionylation was lower in cells transfected with C498A and C670A mutant plasmids than in cells transfected with the wild-type plasmid. However, there was no significant difference in the levels of S-glutathionylation of Pik3r1 in cells transfected with the C656A mutant plasmid. Furthermore, the researcher simultaneously mutated Cys498 and Cys670 and transfected the double-mutant plasmids into Pik3r1-KO RAW264.7 cells. Under the same conditions, Co-immunoprecipitation results showed that the level of GSTP1-mediated S-glutathionylation was completely abolished (Figure 7F), which strongly demonstrated that GSTP1-mediated S-glutathionylation of PIK3R1 during osteoclastogenesis was achieved through Cys498 and Cys670.
To this point, the authors demonstrated through exhaustive in vitro experiments that Cys498 and Cys670 are the two cysteine sites for GSTP1-mediated S-glutathionylation of PIK3R1 and that this redox related modification may regulate autophagic flux and ultimately inhibit osteoclastogenesis through the Pik3r1-AKT-mTOR cascade.
This study explored a completely novel mechanism by which GSTP1 remodels osteoclast related bone homeostasis through a large number of in vitro and in vivo experiments, and for the first time annotated that the cell fate of osteoclasts is determined by a GSTP1-mediated redox-related posttranslational modification autophagy flux cascade axis.
Ubigene can provide in-stock Pik3r1-KO RAW264.7.
Now our KO cell line bank has been expanded to have 3500+ cell lines, as low as $1980 ea, deliver as fast as 1 week. Of which genes includes:
1200+ genes from 26 signaling pathways;
1500+ genes from 27 disease types;
800+ genes from 5 catagories of drug target.
Ubigene also provides express customization as fast as 4 weeks. With comprehensive strategy evaluation, Gene Dependency Assessment System, Ubigene can lead you to a faster, safer, better way for your experiment and research! Check for more service details >>


