How to utilize CRISPR in drug discover (Part 3)--ion channel
Ion channel is a hydrophilic protein microporous channel composed of transmembrane proteins on the cell membrane, which can regulate the difference of ion concentration inside and outside the membrane, and then regulate the cell function. For example, some ion channels help to regulate nerve conduction, myocardial contraction, etc. In addition, there are also ion channels distributed on the membrane of organelles. These channels are on the lysosome and mitochondrial membrane and can control the functions of cell metabolism. 1
Ion channel plays an important role in the human body and are easy to be interfered by drugs. They are important targets for drug development at present. Nowadays, ion channel targeted drugs are widely used to treat arrhythmia, hypertension, local anesthesia, pain, stroke, epilepsy, depression, bipolar disorder, cystic fibrosis, arrhythmia, COPD, autoimmune diseases, diabetes, etc. 2
At present, many studies on ion channels are based on animal level. However, some of them have thoroughly studied the topics related to ion channels, which can be tested by using gene knockout cell lines. Through cell experiments, reliable conclusions can be drawn efficiently. For example, when exploring the role of ion channels in certain stress, cell lines can give researchers a model for more convenient and direct observation. Researchers can use the knockout cell line to conduct stress modeling experiment, and then observe the cell viability or intracellular ion concentration after the knockout of ion channels by comparing with WT cells, so as to infer the importance of ion channels to cell homeostasis. In addition, a more important role of ion channel knockout cell lines is to screen and classify drugs on a large scale. Using the knockout cells of specific ion channels, we can quickly determine the active domain of drugs and quickly and reliably classify drugs that can or cannot affect the activity of ion channels. Of course, knockout cell lines is also a reliable experimental tool when studying the mechanism of ion channels.
In this article, we will introduce a few studies of using ion channel-related genes knockout cell lines.
Knockout TRPV4 can suppress the expression level of IL-8 and relief the low back pain caused by stretching
"Identification of the TRPV4 ion channel as a mechanotransducer and therapeutic target in low back pain" is the study of identifying the TRPV4 ion channel as a mechanical sensor and therapeutic target in low back pain. 3 As a transmembrane protein, TRPV4 belongs to the transient receptor potential Vanilloid (TRPV) family. It can react to temperature, chemicals and mechanical stimuli, and is expressed in a variety of cells and tissues. In addition, TRPV4 can also regulate the intracellular calcium concentration and participate in the process of cell apoptosis, cell proliferation, cell membrane potential regulation and vascular endothelial function regulation. The scientists successfully constructed TRPV4 knockout AF cell line using CRISPR/Cas9 technology, and studied gene expression, release of inflammatory mediators and activation of MAPK pathway. The results showed that knockout of TRPV4 could prevent the expression of IL-8 and IL-6 genes induced by stretching. Therefore, the author provides new evidence that TRPV4 may transmit hyperphysiological mechanical signals to inflammatory signals through p38 in human AF cells. Pharmacological inhibition or gene editing targeting TRPV4 can be used as a potential treatment strategy for discogenic low back pain in patients with fibroblasts injury.
Ubigene has more than 3000 in-stock KO cell lines, such as TRPV4 and genes in 26 signal pathways (MAPK/ERK/AKT/mTOR......), deliver in one week. High-quality homozygous clones as low as $1980! Click here to explore>>
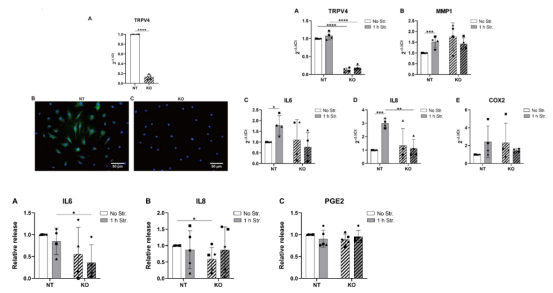
Figure 1. Comparison of relative genes expression in TRPV4-KO cells and WT cells
The effect of Loureirin B on Ca2+ influx and cytokine secretion was studied by KV1.3 knockout cell line
In "Loureirin B exercises its suppressive effects by inhibiting STIM1/Orai1 and KV1.3 channels" 4, the KV1.3 ion channel (KCNA3 gene) was deleted by CRISPR/Cas9 system. And it explores the effect of Loureirin B, an ion channel intervention drug, on Ca2+influx and cytokine secretion. In the immune system, as the potassium ion channel mainly existing on the T cell membrane, KV1.3 channel can regulate the activation state of T cells and regulate the immune response by regulating the intracellular Ca2+concentration. The research team found that knockout KV1.3 in Jurkat cell line can reduce Ca2+influx and IL-2 secretion, but cannot eliminate the influence of Loureirin B.
Ubigene provides gene knockout Jurkat cell line service. With the experience of over 5000 gene editing cases, CRISPR-U™ technology that improves the efficiency of gene editing by 10 times, and the refined cell culture system and differential transfection system, Ubigene can ensure the delivery of high-quality homozygous clones! 200+ cell types can be customized in 4 weeks, click to learn more>>
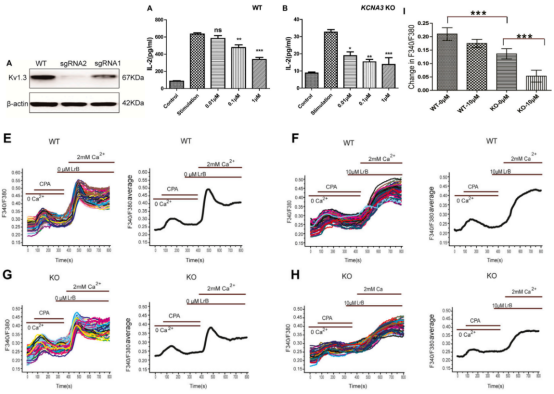
Figure 2. Loureirin B inhibits IL-2 secretion of KCNA3 knockout cells and blocks intracellular Ca2+ reuptake
In the experiment, researchers further proved that Loureirin B can reduce Ca2+ influx and IL-2 secretion by inhibiting STIM1/Orai1. These findings suggest that LrB achieves immunosuppression by inhibiting KV1.3 and STIM1/Orai1 channels.

Figure 3. Loureirin B inhibits STIM1/Orai1 currents in a concentration-dependent manner
CFTR knockout cell line verify the Cl - efflux mediate role of CFTR channel in bone cells
CFTR is an anion channel that regulates the transport of electrolyte and water in epithelial tissue, and its mutation is easy to cause cystic fibrosis. Previous studies have shown that CFTR is also related to mechanical sensitivity. In this regard, "Channel function of CFTR in bone cells for microenvironment homeostasis" 5 has studied the function of CFTR in bone cells, revealed the regulatory mechanism of CFTR on the microenvironment of bone cells, and provided a new strategy for the treatment of osteocyte-related diseases.
The research team found that the intracellular Cl- concentration and depolarization membrane potential of MLO-Y4 cell line increased after treatment with CFTR inhibitors. They speculated that CFTR might mediate the Cl- efflux in bone cells. They successfully constructed the CFTR knockout MLO-Y4 cell line using CRISPR/Cas9 technology. After the treatment with inhibitor, the intracellular Cl - concentration and membrane potential were detected. It was found that the increase of the intracellular Cl - concentration and membrane potential of the CFTR gene knockout cell line was greater, which confirmed that CFTR mediated the Cl - efflux in bone cells. In addition, by comparing the recovery rate of intracellular pH of alkalized MLO-Y4 cells before and after the CFTR knockout, it was found that CFTR can promote the secretion of HCO3 - by cells, suggesting that CFTR can regulate the pH in bone cells by mediating the efflux of HCO3 -, providing a theoretical basis for the treatment of severe osteoporosis and osteomalacia caused by pH changes caused by systemic acidosis.
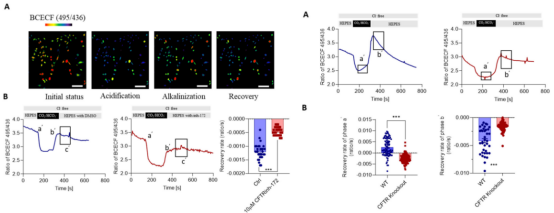
Figure 4. The CFTR knockout MLO-Y4 cell line proves that CFTR can promote Cl - efflux
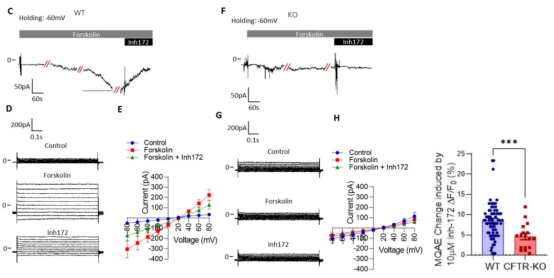
Figure 5. Effect of CFTR inhibitor on HCO3- secretion and pH maintenance of wild-type and CFTR knockout cell line
These are three articles published in the past two years on the mechanism of drug intervention and the mechanism of action of ion channels by using ion channel knockout cell lines. When studying unknown ion channels, researchers are not sure whether the knockout of channel genes will lead to changes in cells interaction, and are willing to spend a long time constructing animal models.
However, we do not have to wait for the long timeline of animal model construction for the well known ion channels. We can use CRISPR/Cas9 technology to knockout or knockin ion channel genes directly on the target cell line for cell-level experiments. Especially for some experiments that require long-term detection of ion channel activity, ion channel knockout cell line is a more ideal model. If the animal model is used, we need to detect the activity of ion channels in the isolated tissues. Even if the brain tissue with relatively strong vitality is put into the maintenance solution, its ideal detection duration is only 4 hours, let alone the more vulnerable myocardial tissue.
In addition, ion channel knockout cell line is the best tool for initial screening of therapeutic drugs. In brief, it has two advantages. The first is that cells have good stability and high repeatability. Compared with animal experiments, cell experiments have lower costs and shorter experimental cycles. Another point is that when conducting cell experiments, we can use human tumor cells as experimental tools, so that we can ensure the homology of the protein structure of the ion channel and improve the reliability of the drug screening results.
Ubigene’s KO cell bank offers several ion channel KO cell lines, such as KCNA3, STIM1, CFTR, etc. Over 3000 homozygous knockout clones deliver in 1 week, only $1980. Click here to explore>>
CRISPR-U™ technology improves gene editing efficiency by 10 times. Ubigene provides EXPRESS custom knockout in 200+ types of cells, as fast as 4 weeks. Talk to our CRISPR expert>>
Reference:
1 Ohya, S., Kito, H., Hatano, N. & Muraki, K. Recent advances in therapeutic strategies that focus on the regulation of ion channel expression. Pharmacol Ther 160, 11-43, doi:10.1016/j.pharmthera.2016.02.001 (2016).
2 Boyden, P. A. & Jeck, C. D. Ion channel function in disease. Cardiovasc Res 29, 312-318 (1995).
3 Cambria, E. Identification of the TRPV4 ion channel as a mechanotransducer and therapeutic target in low back pain, ETH Zurich, (2021).
4 Shi, S. et al. Loureirin B Exerts its Immunosuppressive Effects by Inhibiting STIM1/Orai1 and KV1. 3 Channels. Frontiers in Pharmacology 12, 1298 (2021).
5 Zhang, X. Channel function of CFTR in bone cells for microenvironment homeostasis. (2022).
 Subscribe Us
Subscribe Us Gene Editing Services
Gene Editing Services
 EZ-editor™
EZ-editor™ Red Cotton Gene knockout Project
Red Cotton Gene knockout Project













