How CRISPR/Cas9 facilitates respiratory disease researches?

Respiratory disease is a common and frequently occur disease, including bronchitis, pneumonia, pulmonary tuberculosis, chronic obstructive
pulmonary disease (COPD), lung cancer and nasopharyngeal carcinoma. In
addition, there are other congenital pulmonary disease, such as loss of surfactant protein, cystic fibrosis (CF), α-1 antitrypsin (AAT) deletion, etc. Although these rare respiratory diseases incidence rate is low, it
will have a significant impact on the health and life span of
patients.
Discovering respiratory diseases treatments via CRISPR/Cas9
With the help of CRISPR/Cas9, people have made many breakthrough researches
and applications in respiratory system. In addition to providing
pre-clinical models for the exploration of pathogenic factors and potential
mechanisms of many respiratory diseases, it can also find new disease
drivers or drug targets by screening genes involved in respiratory process,
development and pathology, and even directly edit epigenetic markers leading
to respiratory diseases. Combined with other technologies such as stem cell
reprogramming and transplantation to correct gene mutations, it provides the
possibility for personalized treatment in the future (Fig. 1).
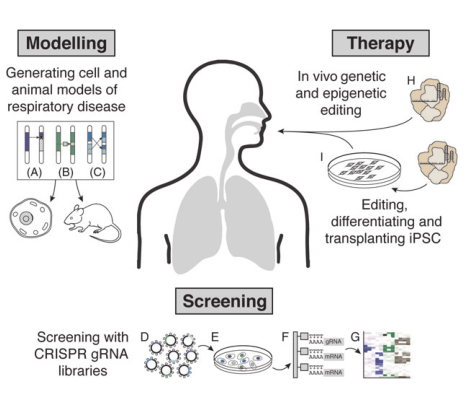
Fig.1 Applications of CRISPR/Cas system in respiratory diseases researches
Case studies
1.Knockout of Grhl2 to study its role in regulating the morphology and physiology of epithelial tissue
The structure, tissue and cells of the airway arranged by pseudostratified
mucosal ciliated epithelium in mammalian lung are very important for many
physiological functions. However, when performing many physiological
functions, lumen cells are vulnerable to viruses, inhaled poisons and
inhaled gastric acid. Fortunately, the loss of lumen cells is usually
compensated by the mobilization of undifferentiated stem/progenitor cells
close to the substrate. In the stable state, these basal cells (BCS) are
relatively static, but when the cells in the cavity are absent, they will
proliferate and differentiate into ciliated and secretory cells. Although
great progress has been made in the regulation of BC cell self-renewal and
offspring lineage selection, the mechanism driving BC cell morphological changes is still unclear. Therefore, through CRISPR/Cas9 system editing technology,
Hogan’s group infected HBE cell line with lentivirus targeting Grhl2 to obtain
Grhl2 knockout cell line, and found that Grhl2 knockout would inhibit organoid morphogenesis and ciliary cell
differentiation, and reduce the expression of notch and ciliary genes
(Mcidas, Rfx2 and Myb) with different Grhl2 regulatory sites. It provides further evidence for the role of Grhl2 in regulating the
morphology and physiological function of epithelial tissue, and also
provides a model for better understanding the function of airway
epithelium.
2.Knockout of Nrf2 to study the protective mechanism of oxidative
stress mediated by oligomeric proanthocyanidin
Oxidative stress can lead to pulmonary fibrosis and respiratory diseases.
Oligomeric proanthocyanidins have been proved to have a protective effect on
hydrogen peroxide induced oxidation in non-small cell lung cancer
cell line (A549) by CRISPR/Cas9 technology. It was found that the expression of Nrf2
changed in A549 cells stimulated by oligomeric proanthocyanidins. Nrf2 is a
transcription factor sensitive to redox reaction, which can cause changes in
its downstream target genes at the transcriptional and protein levels.
Knockout of Nrf2 by CRISPR/Cas9 technology can make Nrf2 unable to bind to
the antioxidant response element ARE, thus eliminating the protective effect of oligomeric proanthocyanidins
against hydrogen peroxide induced oxidative stress. Studies have shown that oligomeric proanthocyanidins play an important role in the antioxidant
response of A549 cells through Nrf2-ARE pathway. This study provides a theoretical basis for the treatment of
respiratory diseases.Ubigene now offer in-stock Nrf2 knockout cell line in A549, THP-1,
RAW264.7 backgrounds, only $1980, deliver in 1 week, click here to learn more >>
3.KO immune checkpoints and intervene growth of lung carcinoma
CRISPR/Cas9 technology has been used as an intervention for lung cancer in clinical
trials. This research mainly studied the function of PD-1. PD-1 can only be
expressed in activated T cells. This gene can promote cell apoptosis and is
a clinical immune checkpoint. Therefore, researchers found that the survival
time of T cells would be prolonged when PD-1 was knocked out, because the T
cell cycle checkpoint was inhibited, resulting in the prolongation of the
survival time of activated T cells. After PD-1 gene was knocked out by CRISPR/Cas9 technology, the number of activated T cells with immune function in blood
increased significantly, so as to inhibit tumor growth. KO cells can not only be used to study the regulation of T cell activity
and enhance its antitumor effect, but also play a good role of negative
screening or negative control in verifying and screening immune
checkpoints. Ubigene’s KO cell bank covers most of the immune checkpoints, click here to explore this collection >>
4.Application of CRISPR/Cas9 technology in the treatment and diagnosis of genetic diseases
CRISPR mediated gene therapy correction in cystic fibrosis (CF) and other
congenital diseases has some advantages over traditional gene
therapy. Because the gene modification induced by CRISPR system is permanently
maintained in edited cells, while the modified gene is still controlled by
its endogenous promoter. Therefore, people pay much attention to the use of
CRISPR/Cas9 technology to correct genetic diseases; In addition, CRISPR system can also
be used as a diagnostic tool. At present, CRISPR system has been used for
the detection of microRNA biomarkers and the enrichment of a small number of
alleles of cell free DNA before the diagnosis and detection of NSCLC
patients. This diagnostic application may be more feasible than in vivo gene
editing or cell transplantation strategies, and may have a direct and
positive impact in the clinical environment.
In conclusion, CRISPR system has surpassed the previously available gene
editing methods and become an important milestone in molecular biology in
this century. The system has been widely used in the construction of cell
and animal models of respiratory diseases and the screening of factors
related to the development of respiratory infection and lung cancer, which
provides promising treatment possibilities for lung and respiratory
diseases. However, the following obstacles must be overcome:
1. gRNA specificity
2. Editing efficiency
3. Target cells and route of administration
4. Vector for gene editing
5. Effects of immunogenicity and DNA damage response on CRISPR
CRISPR-U™, developed by Ubigene, is more than 10X more efficient than traditional CRISPR/Cas9, and greatly improves the success rate of gene
editing. At present, Ubigene has successfully constructed a gene knockout cell bank with nearly 2000 KO cell lines in stock. KO validated gRNA only $20, don’t miss this promotion >>
Reference
[1].Moses, C. and P. Kaur, Applications of CRISPR systems in respiratory health: Entering a new ‘red
pen’ era in genome editing. Respirology, 2019. 24(7): p. 628-637.
[2].Rosen, B.H., et al., Animal and model systems for studying cystic
fibrosis. Journal of Cystic Fibrosis, 2018. 17(2): p. S28-S34.
[3].Maddalo, D., et al., In vivo engineering of oncogenic chromosomal
rearrangements with the CRISPR/Cas9 system. Nature, 2014. 516(7531): p.
423-427.
[4].LiaoX, ZhengSJ, LuKK,et al.Plant polyphenols exert antioxidant
activity of by Nrf2/ARE signaling pathway:areview[J].Shi Pin Ke
Xue,2016,37(7): 227-232.DOI: 10.7506/spkx1002-6630-201607041.
 Subscribe Us
Subscribe Us Gene Editing Services
Gene Editing Services
 EZ-editor™
EZ-editor™ Red Cotton Gene knockout Project
Red Cotton Gene knockout Project









