How to utilize CRISPR in drug
discover (Part 1)--GPCR
Amgen' s erenumab, shown in blue. Image courtesy of Amgen.
Strong light makes people close their eyes, the fragrance of flowers makes people pleasured,
delicious food makes people appetized, and darkness makes people's hearts beat faster... On the surface of
cells, there are a large number of G-protein-coupled receptors (GPCR) that can sense external signals, and they
are involved in regulating all aspects of activities in our bodies.
GPCRs, also known as seven-(pass)-transmembrane domain receptors, have a very conservative
structure. Its N terminus is located outside the cell membrane, and its C terminus is located inside the cell
(Figure 1). Ligands combine with the "pockets" composed of transmembrane helixes to transfer signals to cells
through the change of transmembrane conformation. The nature of the ligand determines the state of GPCR. The
activated ligand (agonist) activates the receptor, and the receptor further couples with the G protein
heterotrimer to transduce the ligand signal into the cell; The inhibitory ligand (inhibitor/antagonist) inhibits
the function of the activated receptor.
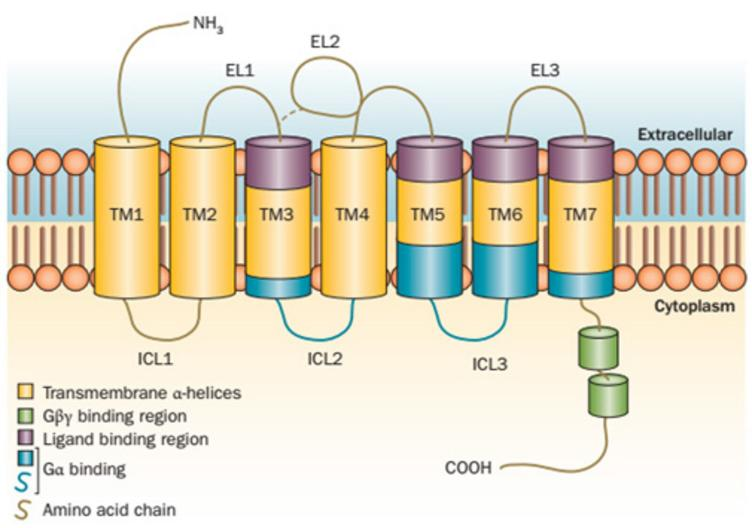
Figure 1. Seven-(pass)-transmembrane structure of GPCR
There are more than 800 GPCR family members in the human body. GPCR family is one of the most
important drug targets nowadays. Cancer, diabetes, heart disease, immune and infectious diseases, neurological
and mental diseases, etc., are related to GPCR. At present, about 1/3 of the drugs approved by FDA are targeting
GPCR, but only about 100 GPCR genes are covered. Apart from the other half of the GPCR genes responsible for
olfaction, there are still about 300 potential targets in GPCR family. It is possible to develop better drugs in
the future through in-depth study and screening of these GPCRs. In 2017, an article on GPCR drug research and
development was published in Nature. It mentioned that there are average of 10.3 drugs targeting each
well-studied GPCR drug target, which means it is necessary to discover new target genes in the
development of new GPCR-related drugs. It is also mentioned that one
of the key problems to be solved in the future R&D of GPCR drugs is the need to improve the disease
models and use gene editing systems such as CRISPR/Cas9[1] to carry out drug target validation. So what is the relationship
between CRISPR/Cas9 and the R&D of GPCR drug targets? In this article, we will introduce some relevant cases
to sort it out.
Validate that GPRC6A is a potential target for developing
prostate cancer antagonists
Prostate cancer is one of the most common cancers and the second leading cause of
cancer-related deaths among American men. Androgen deprivation therapy (ADT) is the initial treatment for this
disease, but the disease usually develops into castration-resistant prostate cancer (CRPC), which is more
clinically invasive. Therefore, it is necessary to further understand the molecular pathogenesis of CRPC and
identify new targets to design new treatments for prostate cancer. GPRC6A is related to the pathogenesis of
prostate cancer, but its role is still unclarified. The researchers used CRISPR/Cas9 technology
to create GPRC6A deficient PC-3 cell line, and compared the changes in cell proliferation
and migration with wild-type PC-3 at the basic level and under ligand stimulation. The results show that GPRC6A knockout PC-3 cells can significantly inhibit the activation of osteocalcin in ERK, AKT, and
mTOR signal pathways, and inhibit cell proliferation and migration in vitro. In vivo xenotransplantation
model, compared with wild-type PC-3 cells, GPRC6A knockout cells had significantly reduced tumor growth,
indicating that they were resistant to the growth of prostate cancer induced by osteocalcin. The research results support the role of GPRC6A in prostate cancer and suggest a potential
therapeutic target to inhibit prostate cancer [2].
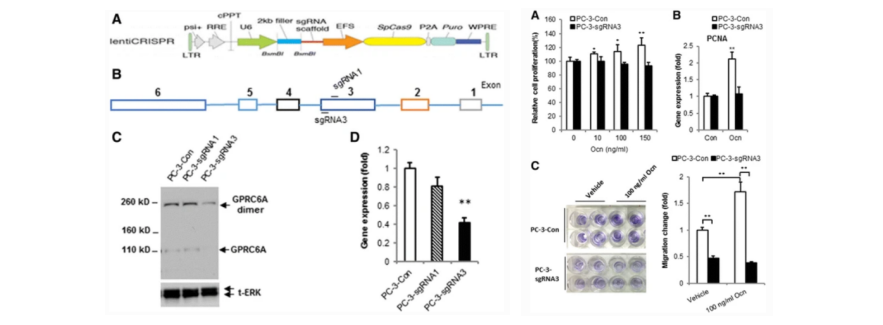
Figure 2. CRISPR/Cas9 mediated GPRC6A knockout in
PC-3 cell line construction and its proliferation and migration ability detection
The exclusive KO cell bank of Ubigene provides in-stock KO cell lines of 25
signal pathways, such as ERK, AKT and mOTR, covering thousands of genes, as low as $1780, and homozygous KO clones are delivered in 1
week! Click here to
explore your target genes>>
Validate the activity of different receptors of GIPR/GLP-1R
dual agonists by gene editing
After the intake of nutrients, L cells and K cells in the intestine secrete Glucagon-like
peptide-1 (GLP-1) and Glucose-dependent insulinotropic polypeptide (GIP), respectively. These incretins can
stimulate insulin secretion by increasing the glucose of pancreatic βcells (GSIS) to
reduce the blood glucose level. This has established the receptors GLP-1R and GIPR as important drug targets for
diabetes treatment. Research shows that the combined use of GLP-1 and GIP can produce beneficial effects.
However, due to the overlap of downstream signal pathways of the two receptors, it is necessary to determine the
activity of dual agonists on each receptor when developing potential therapies. The researchers
successfully obtained GIPR-KO and GLP1R-KO clones by targeting the GIP receptor (GIPR) and
GLP-1 receptor (GLP1R) respectively with CRISPR/Cas9 technology in the INS-1 pancreatic β cell line. The obtained cell lines were used to test the activity of
different dual agonists. Dual agonist A has been found to be quite effective in wild-type INS-1 cells and
GIPR-KO cells, but its efficacy in GLP1R-KO cells is 20 times lower, indicating that GLP-1 contributes more to
dual agonist A, while dual agonist B results showed that GIP contributes more. Balanced dual agonist C
participates in GLP-1R and GIPR in vitro and in vivo. This is the first time that CRISPR/Cas9
technology has been used to detect the activity of GLP-1R/GIPR dual agonists in
vivo. In addition, this technology can also be used to study many other co-expressed
targets in βcells and other related cell lines [3].
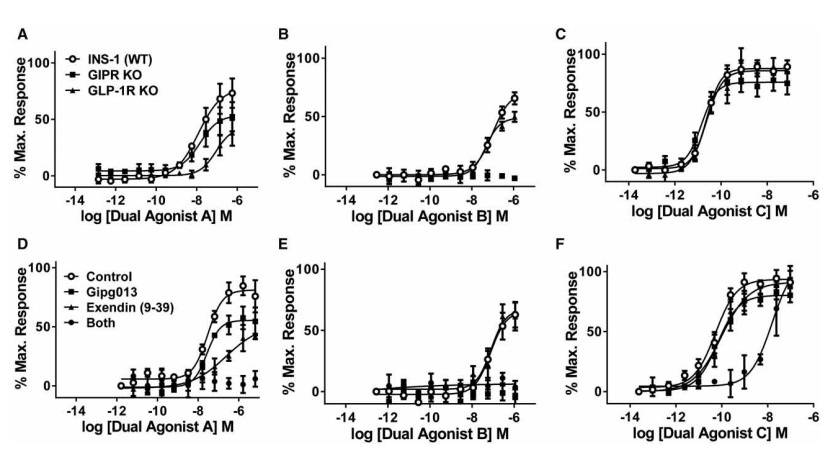
Figure 3. Tests of GIPR or GLP-1R
components in dual agonists
Validate the classical β2AR target by
genetic method to supplement the pharmacological evidence of this drug target
βAdrenergic Receptor (βAR) is a G-protein-coupled
receptor, which is critical to the physiological response of hormone/neurotransmitter adrenaline and
norepinephrine found in the nervous system and the whole body. It is the target of many widely used drugs, among
which βAR and β2AR are the most
widely studied, and their ligands are used for the treatment of asthma and cardiovascular diseases. βARs send signals through Gαs G protein and by activating
adenylate cyclase and cAMP-dependent protein kinase. In order to supplement pharmacological evidence for the
role of β2AR in the Ca2+ release process, researchers have studied it at the genetic level. Create
HEK293 cells with ADRB2 gene deletion through CRISPR/Cas9 system, transfect the knockout lines with control
vectors or HA-β2AR expressing constructs,
and test their Ca2+ response to ISO. The
results show that in the absence of β2AR, no Ca2+ response to ISO can be detected in cells, and only very weak non-specific response
can be observed at much higher concentrations. Transfected with the β2AR expression plasmid can rescue this
situation [4].
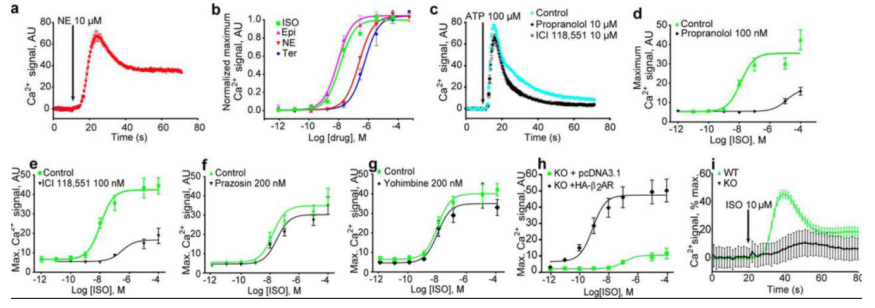
Figure 4. Tests of
Ca2+ signal in HEK293 cytoplasm
There are many cases in which CRISPR/Cas9 technology has been
applied to GPCR research, such as negative screening of genes promoting viral infection through
the sgRNA knockout library of GPCR, and labeling of interested GPCR genes
through CRISPR/Cas9 technology. Horioka et al. [5] knocked LgBiT (an 18 kDa protein), a fragment of NLuc, into the N-terminal of CXCR4 gene in
HEK293 cells, and at the same time knocked SmBiT (an 11 amino acid residue peptide), another segment of NLuc,
into the C-terminal of β- arrestin2. When these two proteins are close, fluorescence
can be emitted, which is used to screen agonist ligands of CXCR4.
GPCR family still has many “secrets” that have not been explored. It
has great potential in drug research and development and is expected to provide hope for the treatments of major
diseases. With the development of CRISPR/Cas9 technology, we can use this method to generate cell models to study
GPCR, validate and screen drug targets, and accelerate the development of GPCR-targeting drugs.
There are more than 100 kinds of GPCR-KO cell
lines in Ubigene’s KO cell bank, visit us to find out the list >>
In addition, Ubigene provides customized KO service as fast as 4 weeks, GPCR
expression stable cell line construction service as low as $2020, which brings you an
extremely fast and high-quality construction experience and assists in high-throughput screening of drug
targets! Click and contact us to learn more about the services>>
Reference:
[1] Hauser A S, Attwood M M, Rask-Andersen M, et al. Trends in GPCR drug discovery: new
agents, targets and indications[J]. Nature reviews Drug discovery, 2017, 16(12): 829-842.
[2] Ye R, Pi M, Cox J V, et al. CRISPR/Cas9 targeting of GPRC6A suppresses prostate
cancer tumorigenesis in a human xenograft model[J]. Journal of Experimental & Clinical Cancer Research, 2017,
36(1): 1-13.
[3] Naylor J, Suckow A T, Seth A, et al. Use of CRISPR/Cas9-engineered INS-1 pancreatic β cells to define the pharmacology of dual GIPR/GLP-1R agonists[J]. Biochemical Journal,
2016, 473(18): 2881-2891.
[4]Galaz-Montoya M, Wright S J, Rodriguez G J, et al. β2-Adrenergic receptor activation mobilizes intracellular calcium via a
non-canonical cAMP-independent signaling pathway[J]. Journal of Biological Chemistry, 2017,
292(24): 9967-9974.
[5]Horioka M, Huber T, Sakmar T P. Playing Tag with Your Favorite GPCR Using CRISPR[J]. Cell
Chemical Biology, 2020, 27(6): 642-644.
 Subscribe Us
Subscribe Us Gene Editing Services
Gene Editing Services
 EZ-editor™
EZ-editor™ Red Cotton Gene knockout Project
Red Cotton Gene knockout Project












