[Research highlight] Noninvasive detection of ACE2 expression
Background
The outbreak of coronavirus disease 2019 (COVID-19) at the end of 2019 led to a global pandemic caused by severe acute respiratory syndrome coronavirus type 2 (SARS-CoV-2). It was reported that low-dose radiation reduced inflammation and promoted healing of COVID-19. Angiotensin-converting enzyme 2 (ACE2) is a homologue of carboxypeptidase ACE, and the main active peptide of the renin-angiotensin system (RAS). The spike protein of SARS-CoV-2 interacts with ACE2 of human respiratory epithelial cells, which leads to infection. The development of inhibitor probes targeting ACE2 is more reasonable in theory, and more possible in practice than antibody drugs targeting the virus itself. The receptor binding domain (RBD) is the intermediate between virus and ACE2 receptor. Some researchers proposed that the modification of RBD with PET radionuclide may give some characters of the SARS-CoV-2’s deadly entry.
Using advanced PET technology to monitor the labeled RBDs metabolism and residual may provide a comprehensive viewpoint of SARS-CoV-2 s infections in animal. Therefore, the researchers from Key Laboratory of Tumorigenesis and Transformation of Beijing Ministry of Education, Dan Li et al. published an article named “SARS-CoV-2 receptor binding domain radio-probe: a non-invasive approach for angiotensin-converting enzyme 2 mapping in mice” in Acta Pharmacologica Sinica. They constructed a radioiodinated RBD as a molecule-targeted probe to non-invasively explore ACE2 expression in vivo, and to investigate radiotherapy pathway for inhibiting ACE2.

The high affinity and selectivity of 124I-RBD antibody against ACE2
RBD was labeled with [124I]NaI using an N-bromosuccinimide (NBS)-mediated method, and 124I-RBD was obtained after purification with a specific activity of 28.9 GBq/nmol. Its radiochemical purity (RCP) was over 90% in saline for 5 days. The binding ability of RBD to human ACE2 receptor was studied by SPR binding studies. Affinity model was applied to fit the data and to calculate the dissociation constants, KD = 14.08 nM (Fig. 1). The binding potency of 124I-RBD to human ACE2 was detected by the ELISA method, with the dissociation constants, KD =75.7 nM (Fig. 2). The results showed that RBD has a high affinity with hACE2 in vitro.
Compared with non-transduced HeLa cell line, the ACE2 gene expression rate of HeLaACE2+ cell line (provided by Ubigene) was 561.2 times higher. Western blot analysis showed that HeLaACE2+ cell did have high expression of ACE2. Cell uptakes of radiotracers are summarized in Fig. 3. Results showed that at 2h, the uptake values of 124I-RBD in HeLaACE2+ cells were (2.96±0.35)% ID/106 cells without blocking and (1.71±0.23)%ID/106 cells with cold RBD (P = 0.00038); the uptake values of 124I-RBD in HeLa cells were (1.96±0.22)%ID/106 cells without blocking and (1.64±0.15)%ID/106 cells with cold RBD (P > 0.05) (Fig. 3).
In this study, Ubigene provided ACE2 overexpression HepG2 and HeLa cell lines to verify the affinity between RBD and ACE2 at the cellular level. Relying on rich cell culture experience, Ubigene has a comprehensive database of the optimal MOI of each cell line. And we offer customized overexpression or knockdown strategies for your particular project. To learn more of our stable cell line services>>

Fig. 1 SPR Response Units [RU] at equilibrium as a function of the concentration of RBD, using immobilized ACE2 as receptor
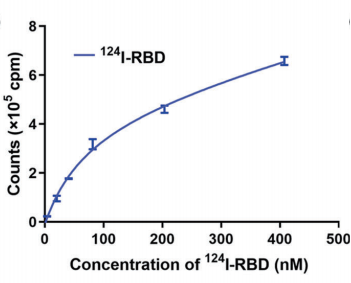
Fig. 2 Binding affifinity assay of 124I-RBD to ACE2.
Fig. 3 The cellular uptake of 124I-RBD in HeLa and HeLa ACE2+ cells respectively. ns represent no statistically significant difference, *P < 0.05, **P < 0.01, ***P < 0.001.
Specific binding of RBD to ACE2 lead to the retention of 124I-RBD in lung
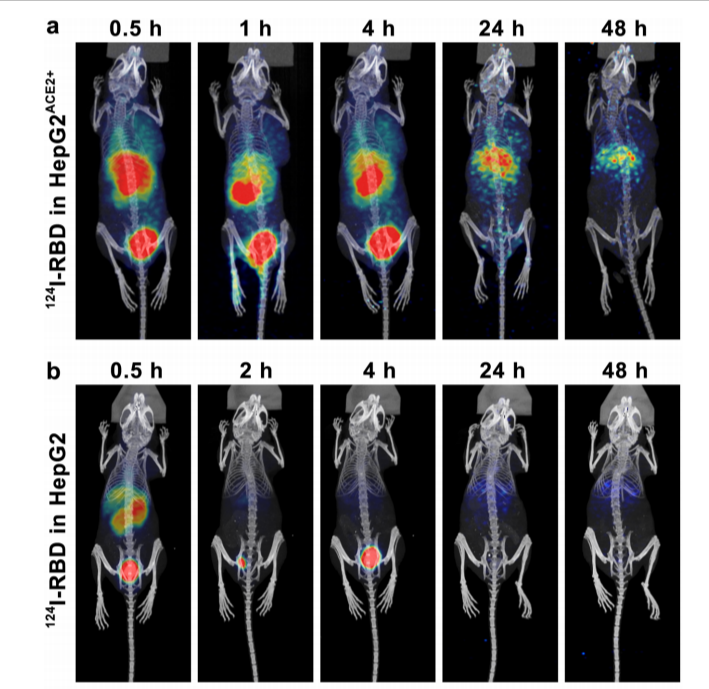
In order to study the retention of RBD in the lung, they adopted the method of intrapulmonary transbronchial administration, and injected 0.927 MBq 124I-RBD in a volume of about 50μL. The micro-PET imaging results were shown in Fig. 4. After intrapulmonary injection, PET/CT imaging showed that the lung retained a long period of high uptake for at least 4 h after the injection of 124I-RBD, and the SUVmean value of the lung was still high for 24 h. At 0.5 h p.i., intrapulmonarily injected [124I]NaI could be observed in the lung, but the SUVmean value in the lung decreased to very low level at 1 h, indicating the specific interaction(Fig.4). Compared to nude mice, mice bearing HepG2 tumors (HepG2ACE2+) showed higher uptake in tumor after 124I-RBD was injected through tail vein. At half an hour, the HepG2ACE2+ tumor site begins to uptake 124I-RBD, the concentration reaches a peak at 1 h, then gradually decreases. At 4 h, it still had a high uptake at the tumor site(Fig.5). As a proof of concept, the researchers conclude the retention of 124I-RBD in the lung is caused by the specific binding of RBD to ACE2, and it is useful for non-invasive mapping in body.
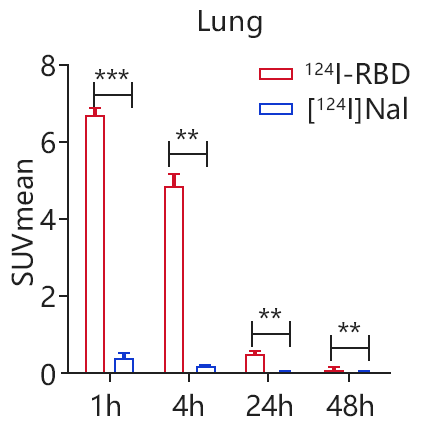
Fig. 4 The SUV changes of 124I-RBD in lung as well as the control [124I]NaI at different time points.
Fig. 5 Micro-PET imaging of tumor models.
In this study, the researchers successfully constructed 124I-RBD, for the first time, as a novel molecular targeting probe for COVID-19. The probing of ACE2 expression via radiolabeled RBD can be used for non-invasive ACE2 mapping. In combination with the available literature, it can be suggested that human-ACE2 targeting monitoring is feasible using iodine isotope modified RBD, which supplies preclinical experience for radiotherapy.
Ubigene is an enterprise focusing on gene editing cell line. We provide high-quality gene-editing cell lines, stable cell lines, virus packaging and other related services and products, as well as nearly 2000 KO cell lines. Please contact us for more details!
 Subscribe Us
Subscribe Us Gene Editing Services
Gene Editing Services
 EZ-editor™
EZ-editor™ Red Cotton Gene knockout Project
Red Cotton Gene knockout Project